|
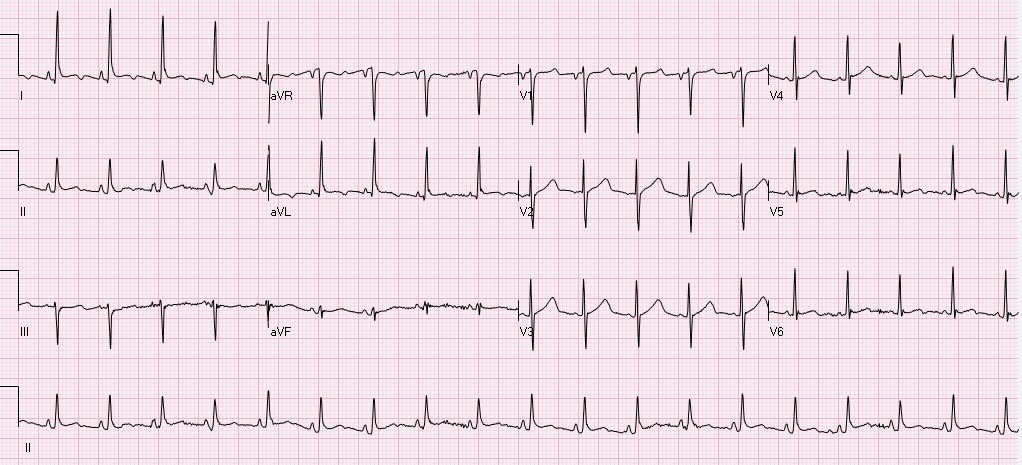
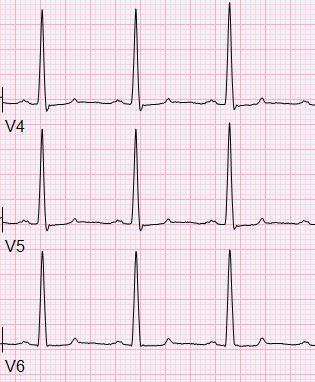
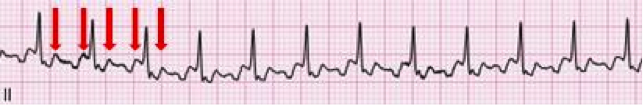
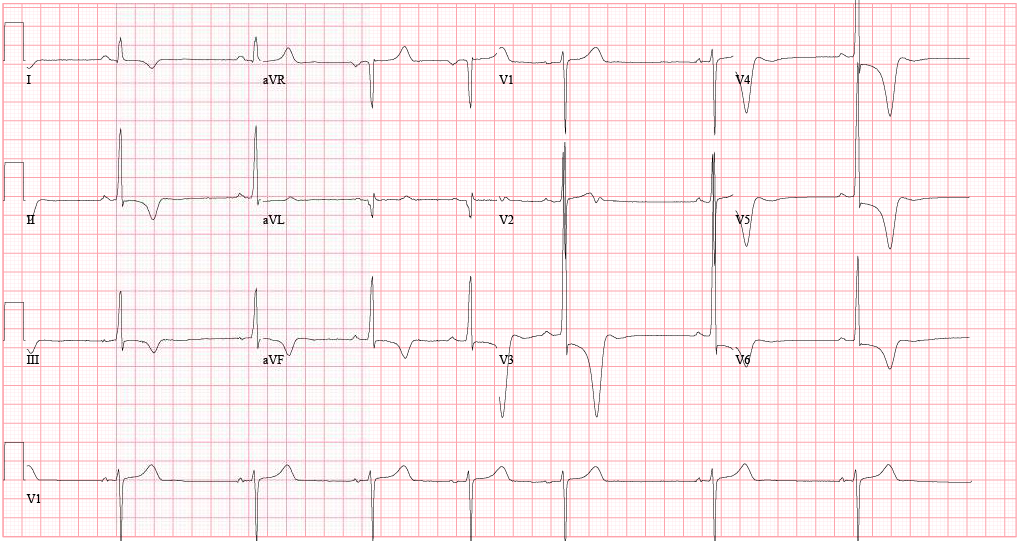
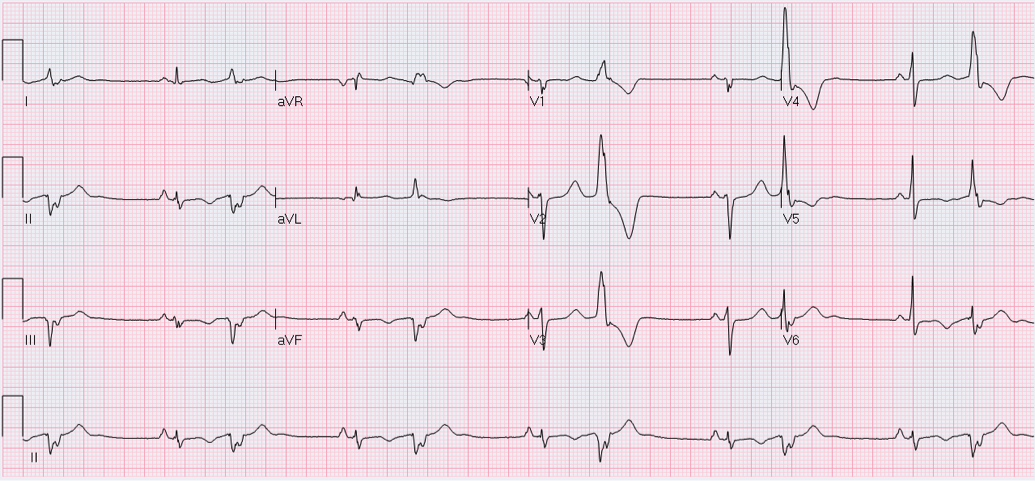
HPI: 50-year-old African-American female with PMH of untreated hypertension, CKD secondary to membranous glomerulonephritis, possible schizophrenia, ongoing cocaine abuse (last use 3 days prior to admission), admitted for treatment of CAP. Chest CT showed bilateral lower lobe infiltrates with moderate pericardial effusion and small bilateral pleural effusions. Cardiology consulted for concern for pericardial tamponade, as primary team reported continued shortness of breath and findings of pulsus paradoxsus and electrical alternans on EKG. EKG: Questions: What does cardiac tamponade look like on an EKG? Does this EKG exhibit electrical alternans and confirm the presence of cardiac tamponade? How sensitive/specific is EKG in detecting cardiac tamponade? EKG Interpretation: Sinus tachycardia with a rate of 114. Horizontal axis. Normal voltage. Borderline LVH. Nonspecific T wave abnormalities, no significant ST depression or elevation. Artifact consistent with respiratory variation. Discussion: EKG findings and Sensitivity/Specificity for cardiac tamponade: • Sinus tachycardia o Sensitivity 76%, Specificity 60% 1 o OR: 4.9 (95% CI: 2.22-10.80) • Low QRS voltage o Sensitivity 56%, Specificity 74% 1 o OR: 3.7 (95% confidence interval [CI]: 1.65-8.30) • Electrical alternans o Sensitivity 23%, Specificity 98% 1 o OR: 12.3 (95% CI: 1.58-95.17) o Electrical alternans is characterized by a cyclical, beat-to-beat shift in the QRS axis, caused by the heart swinging back and forth like a pendulum in a large pericardial effusion relative to the stationary EKG leads. o Video: http://www.nejm.org/doi/full/10.1056/NEJMicm1408805 o This is in comparison to this patient’s EKG, which shows normal respiratory variation over several beats, more notably in her V1 rhythm strip below. These are more pronounced in this patient as she was dyspneic and tachypneic given her pulmonary disease. o Normal respiratory variation: • Presence of all 3 (Sensitivity 8%, Specificity 100%) 1
Does our patient have cardiac tamponade? • EKG does reveal sinus tachycardia, but not electrical alternans or low voltage. We relied on echocardiogram and clinical presentation to confirm that the patient did not, in fact, have cardiac tamponade. Pro-tip: • Do not rely on EKG alone for diagnosis of or screening for cardiac tamponade. • Do not confuse respiratory variation with electrical alternans on EKG. By: Dean Tanner, PGY1 Resources: 1. Role of a 12-lead electrocardiogram in the diagnosis of cardiac tamponade as diagnosed by transthoracic echocardiography in patients with malignant pericardial effusion. Argula, RG, et al. Clin Cardiol. 2015 Mar;38(3):139-44. doi: 10.1002/clc.22370. Epub 2015 Feb 18. 2. Electrical Alternans with Pericardial Tamponade. Jehangir, W, Osman, M. N Engl J Med 2015; 373:e10August 20, 2015DOI: 10.1056/NEJMicm1408805
0 Comments
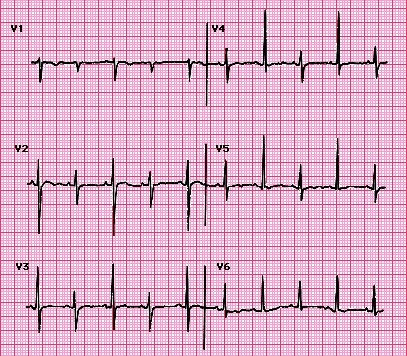
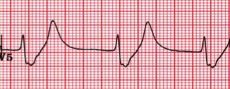
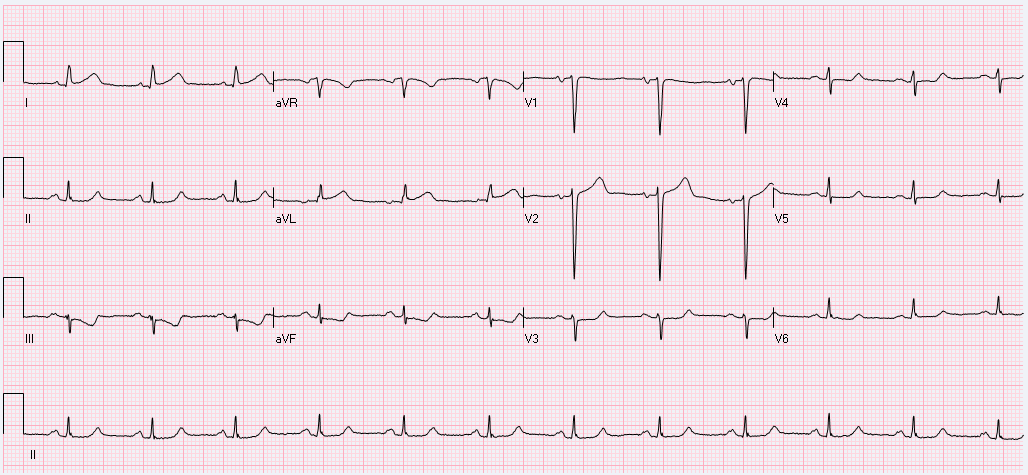
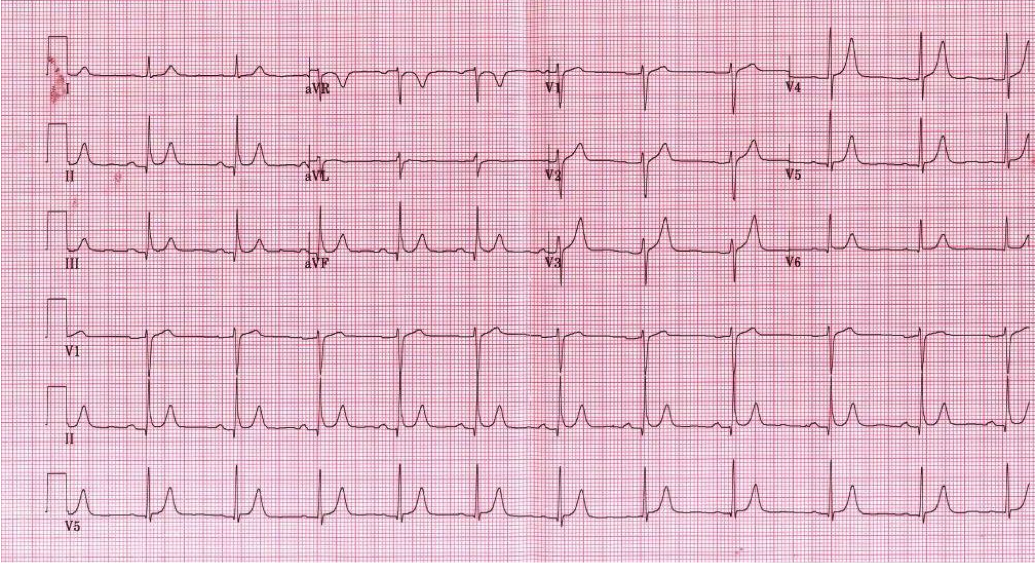
HPI: Patient is a 50 yo F with a history of ESRD s/p failed renal transplant now on hemodialysis who had onset of GI bleeding with associated lightheadedness and hypotension. An EKG was obtained. EKG: Question: What finding on this EKG warrants further management? EKG Interpretation: Normal sinus rhythm at a rate of 77, normal PR interval, normal QRS, normal axis, LVH (significantly increased voltages), narrow based, peaked T-waves, T-wave inversions in leads III and aVF. Discussion: The narrow based, peaked T-waves present in this patient’s EKG, most evident in leads V3-V4, indicate probable hyperkalemia in this patient who is at risk for elevated potassium given her ESRD. The T-wave abnormalities in this EKG are somewhat subtle, but the narrow, “pinched down” bases of the T-waves should warn you of hyperkalemia. This patient’s potassium level was found to be 6.9 and was appropriately treated. Pro-tip:
Potassium can be the great imitator on EKGs because it can cause a wide variety of abnormal EKG findings, including (but not limited to): • Peaked T-waves (often the earliest EKG sign of hyperkalemia) • Prolonged PR interval • Loss of P waves • AV block • Broad QRS complexes with abnormal morphology • Sine waves (usually with extremely severe hyperkalemia) Management:
For patients with ESRD on hemodialysis, dialysis is the definitive treatment for hyperkalemia. Typical management of hyperkalemia (for K>6.0 or EKG changes) includes**: • Calcium (either IV calcium gluconate or calcium chloride), which stabilizes cellular membranes by antagonizing the effect of potassium on membrane potential. Usually reserved for K>7.0 or EKG changes. • Insulin to promote the movement of potassium into cells. • Albuterol and Sodium bicarbonate can also be used to promote the movement of potassium into cells depending on potassium levels. • Kayexalate (Sodium polystyrene sulfonate) can be used to promote potassium elimination from the body, however this has no role in the acute management of hyperkalemia with EKG changes. **Caution in patients with DKA because total body potassium may actually be low and aggressive management of hyperkalemia can result in hypokalemia** By: Krystin Thomas, PGY1 Resources: • Stone, CK. Fluid, Electrolyte, and Acid-Base Emergencies. In Current diagnosis & treatment emergency medicine. 6th ed. New York: McGraw-Hill; 2008. • Burns, E. Hyperkalemia [Web log post] Retrieved August 28, 2015, from http://lifeinthefastlane.com/ecg-library/basics/hyperkalaemia/ HPI: 90 y/o woman h/o sick sinus syndrome, AF, and a pacemaker presents with atypical CP. Troponins are negative, CTPA is negative for PE. She denies palpitations, shock-like sensations, or chest wall contractions. Palpated HR corresponds with QRS complexes on the monitor. EKG: Question:
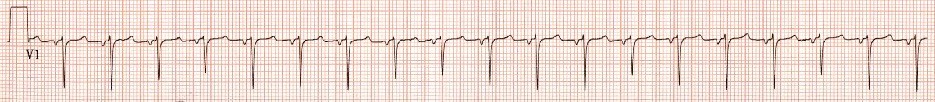
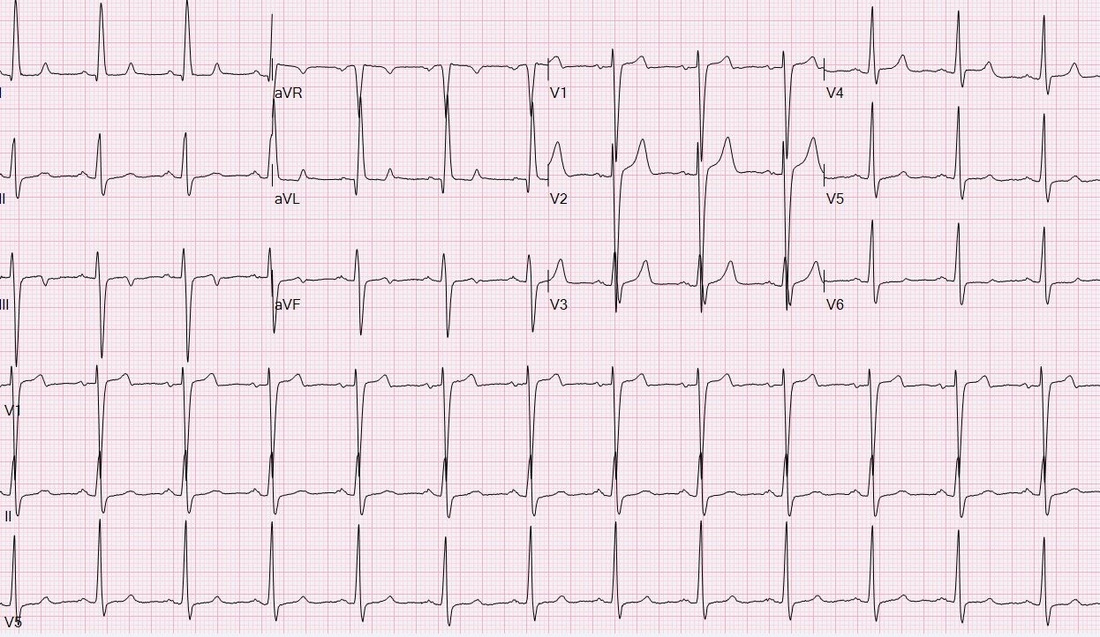
What's going on with this pacemaker? Is the malfunction the cause of her symptoms? EKG interpretation: Ventricular paced rhythm at a rate of approximately 70. QRS complexes are wide. QRS in V1-V6 are predominantly downgoing as are II, III, and aVF consistent with RV pacing. P waves with variable PR interval are seen in II, III, aVL, aVF, and some of the V leads. Pacer spikes are inconsistently seen in I, II, V1-V3. Discussion: This patient presents with atypical chest pain and an apparent pacemaker malfunction. Her pulse corresponds to her QRS complexes, so what's actually going on? As it turns out, this patient is being paced appropriately at her predetermined rate. Interrogation of her PM revealed normal functionality including normal lead impedance (high impedance can signify a fractured PM wire, low impedance can signify cracked insulation). The issue with the EKG here is that some 12 lead machines pick up small pacer spikes in the surface EKG and amplify them so they are visible to the provider. Unfortunately, some machines will only intermittently detect and amplify these spikes due to their signal processing and occasionally will pick up on interference and amplify it as a pacer spike. If you look closely at aVL, she has tiny pacer spikes before her QRS that the machine did not amplify. In this case, the machine used to record the EKG was a GE Marquette MAC 5000. Note that the QRS complexes in the V1 rhythm strip are all identical despite the intermittent pacer spikes suggesting that the pacemaker is actually generating the impulse for each QRS. Management: - Treat the patient as you normally would... labs, CTPA, ASA, NTG, etc as appropriate - If PM malfunction is suspected, interrogate the device. - Consider machine artifact in a patient with "intermittent" pacing who has consistent QRS complexes on their EKG and an regular HR - CXR can be very useful for identifying pacemaker type and manufacturer; focus on the identification tag on the device By: Dr. Andrew Godfrey, PGY1 References: 1. Butscek R, Farrell RM, Littmann. ECG quiz: what is the cause of the apparent pacemaker malfunction? J Electrocardiol. 2013 Mar-Apr;46(2):108-9. 2. Littmann L, Farrell RM. Potential misinterpretations related to artificial pacemaker signals generated by electrocardiographs. J Electrocardiol. 2015 Jul-Aug;48(4):717-20 HPI: 50 y/o male with hx of COPD, heroin use, and recently diagnosed cor pulmonale presents with 3 weeks of lower extremity edema. No chest pain, shortness of breath, or palpitations. An EKG is obtained in the emergency department. Question: What does this EKG tell you about the etiology of this patient’s lower extremity edema? EKG Interpretation: 2:1 atrial flutter with a rapid ventricular response at a rate of 156, narrow QRS (may appear wide due to buried P-waves), right axis deviation, RBBB. Discussion: The patient presents in 2:1 atrial flutter with a ventricular rate of 156, meaning that the atria are firing at a rate 2 times faster (approximately 312). The extra P-waves give the EKG baseline the typical “sawtooth” appearance of atrial flutter. Typical characteristics of atrial flutter:
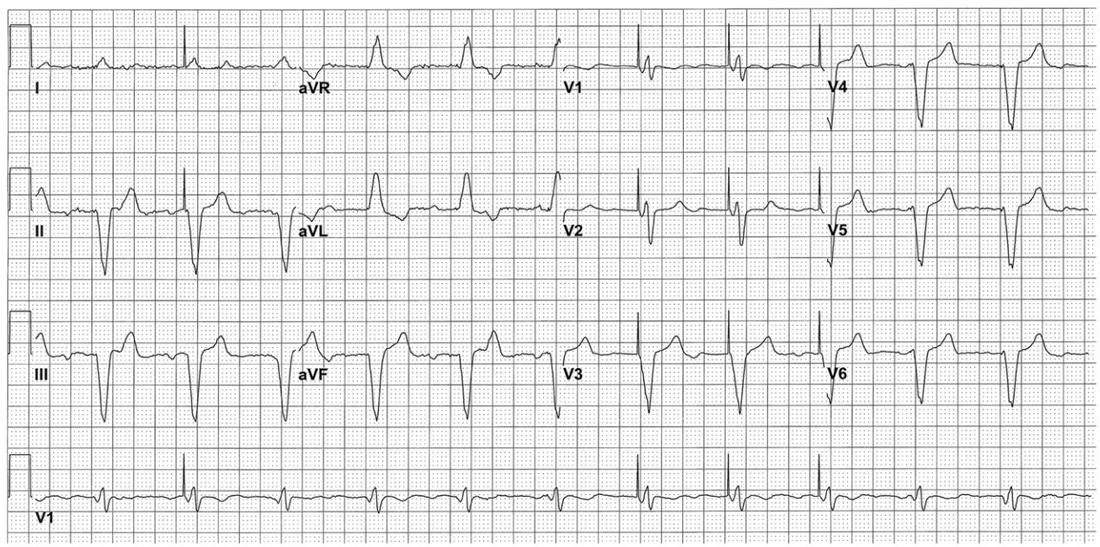
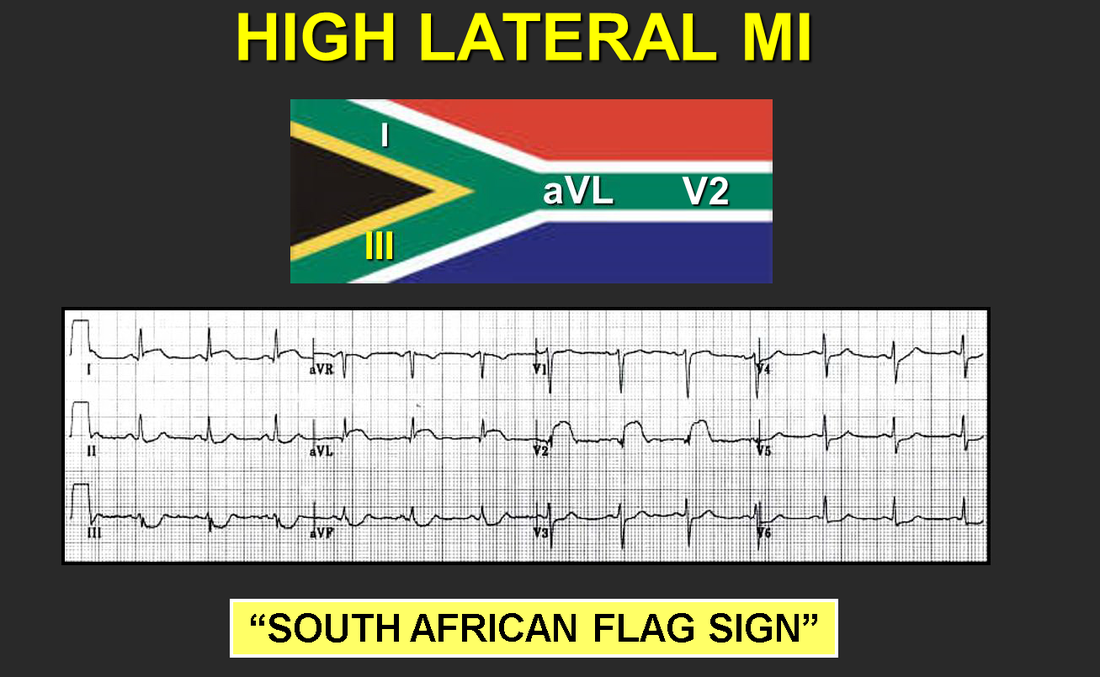
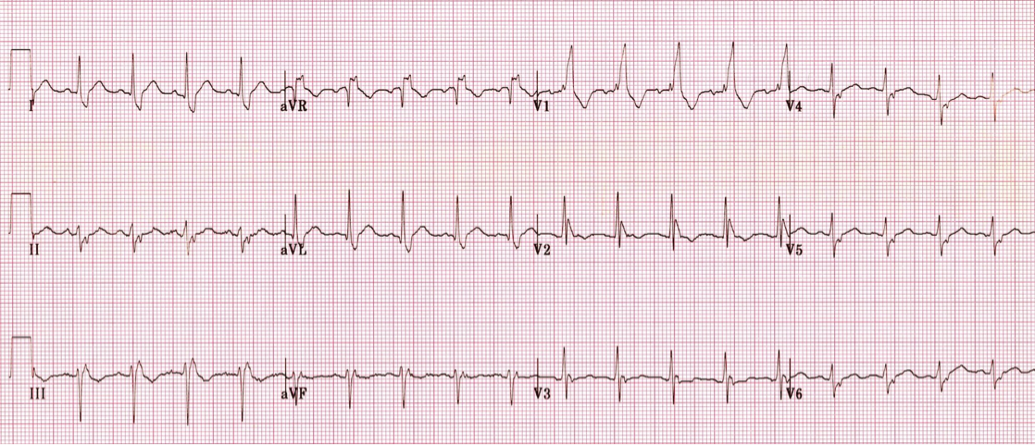
· Regular atrial rate – between 250-350 bpm · Sawtooth flutter waves – directed superiorly and most visible in leads II, III, and aVF · Atrioventricular block – usually at 2:1, but occasionally greater, associated with ventricular rate between 125-175 bpm Pro-tip: If you ever have difficulty distinguishing the buried P-waves of atrial flutter, you can block down the AV node with vagal maneuvers or Adenosine and the slowing of the ventricular rate will unmask the underlying atrial rhythm. Narrow-complex tachycardia at a rate of 130-170: suspect flutter!! Atrial flutter was the cause of this patient’s severe lower extremity edema extending to the scrotum and pre-sacral level. It is not uncommon for patients in atrial flutter to present with edema due the lack of forward flow through the heart secondary to atrial flutter. This patient’s atrial flutter is most likely a result of his severe cor pulmonale, his right atria and ventricle were found to be massively dilated on echocardiogram, predisposing patient to atrial flutter. In general, patients with right heart disease are more likely to get atrial flutter rather than atrial fibrillation, however “where there’s fib, there’s flutter” and these patients can exhibit episodes of a.fib as well. Management: If the patient is stable, the first priority is to achieve ventricular rate control with beta-blockers or nondihydropyridine calcium channel blockers. This patient was given a Diltiazem bolus and then placed on a drip which improved his rate into the 80s, although he was still in 2:1 atrial flutter. Long-term management options for stable patients can be divided into rate control or rhythm control. Rhythm control options: · Cardioversion is an option if patient is known to have been in this rhythm for < 48 hours, if > 48 hours patient requires either: a TEE without evidence of mural thrombus + Heparin or >= 3 weeks of anticoagulation prior to cardioversion (all patients need 4 weeks of anticoagulation post-cardioversion as well) · Catheter ablation for select patient’s (larger right atrium size correlates with lower success rates) Rate control options: · Calcium channel blockers · Beta-blockers By: Dr. Krystin Thomas, MD References: 1. Tintinalli JE. Cardiac Rhythm Disturbances. In: Tintinalli's Emergency Medicine a Comprehensive Study Guide. 7th ed. New York: McGraw-Hill; 2011. 2. Burns, E. Atrial Flutter [Web log post] Retrieved August 15, 2015, from http://lifeinthefastlane.com/ecg-library/atrial-flutter/ HPI: 57 y/o woman with remote hx of CABG/PCI presents with 2 hrs of 6-10/10 substernal chest tightness and SOB. No recent cardiac events/complaints. Initial troponin is 1.48. EKG Interpretation: normal sinus rhythm at a rate of 71 with a normal axis. Narrow QRS complexes throughout, normal intervals. Mild ST elevation in I, aVL, and V2. Inverted T wave in III with mild ST depression as well. No previous EKG available for comparison. Discussion: Although this patient does not have an obvious STEMI on their EKG, there is mild elevation in I, aVL and V2 with reciprocal changes in III, a pattern similar to the South African flag. Concern should be high for a high lateral MI which is an area of that is poorly represented by standard EKG lead placement. In this case, the patient had an acute thrombotic occlusion of a diagonal branch of the LAD artery. On the initial EKG, the patient has lower voltages in their limb leads as well as V3-V6. Although they don’t have frank ST elevation ≥ 1 mm in any lead other than V2, their mild ST elevation should be concerning given the low(er) QRS voltage. Move one of the V leads higher into the left axilla (2-3 interspaces) to obtain a tracing of the high lateral wall. Make sure to discuss with Cardiology your concern for a potential STEMI despite the lack of qualifying ST elevation.
Management: - Obtain EKG tracing of high lateral wall - Labs including troponins as well as CXR - MONA, heparin - Cardiology consult emphasizing the concern for ST changes in the setting of low QRS voltage - Consider emergent cath for patients with persistent chest pain and persistent ST changes By: Dr. Andrew Godfrey HPI: Elderly male with history of HTN, DM, and early dementia presents to ED following a witnessed syncopal event at home. Family denies seizure activity, trauma, or anticoagulation. He is now asymptomatic and back at clinical baseline. Laboratory workup and CXR are unremarkable. EKG is shown below: ECG: ECG Interpretation: Normal sinus rhythm w/ bifascicular block (RBBB + LAFB). Unchanged from multiple old ECGs dating back more than 8 years. Discussion: Bifascicular block is a combination of Right bundle branch block (RBBB) with either Left anterior fascicular block (LAFB) or Left posterior fascicular block (LPFB). When present, ventricular conduction is dependent on only the single remaining fascicle. EKG findings: · RBBB, and either · Left axis deviation > -45o, or · Right axis deviation > +110o Bifascicular block is a common benign finding. It is indicative of extensive conducting system disease, but risk of progressing to complete heart block is minimal (<1% per year). In isolation, no acute treatment is indicated. HOWEVER, syncope is a red flag! Despite no change from old ECGs, in a patient with known bifascicular block a new finding of syncope can indicate intermittent 3rd degree AV block. Patient is at high risk for PEA arrest, ventricular standstill, and sudden cardiac death. Management:
· Admit to telemetry-monitored floor · Mandatory cardiology consultation · Likely will receive permanent implanted pacemaker By Dr. Blake Johnson References: 1. McAnulty JH, Rahimtoola SH, Murphy E, DeMots H, Ritzmann L, Kanarek PE, et al. Natural history of “high-risk” bundle-branch block: final report of a prospective study. N Engl J Med. 1982 Jul 15; 307(3):137-43. PMID: 7088050 2. Burns, E. Bifascicular Block [Web log post]. Retrieved July 28, 2015, from http://lifeinthefastlane.com/ecg-library/basics/bifascicular-block/ HPI: 20 y/o otherwise healthy male presents after syncopal event while playing basketball. He is athletic, plays basketball 7 days a week, and has never syncopized before. There was a prodromal sensation of “lightheadedness”, but he denies other symptoms and is asymptomatic upon ED arrival. EKG is obtained immediately: EKG: EKG Interpretation:
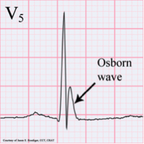
Sinus bradycardia (46 bpm) w/ profound LVH and repolarization abnormality, diffuse ST depression and T-wave inversions. No prior EKG to compare. Discussion: Large negative T-waves have a broad DDx, including ACS, cocaine toxicity, CNS disorders, metabolic abnormalities, etc. In addition, LVH is a common incidental finding on EKG as well. In a young healthy patient without HTN or aortic stenosis, LVH should raise clinical suspicion for Hypertrophic Cardiomyopathy (HC). Additionally, large deep symmetrical T-wave inversions are typical for the Apical Variant of HC (only 3-5% of total HC cases). Importantly, since there is no LV outflow tract obstruction in this variant, the classic murmur associated with Hypertrophic Obstructive Cardiomyopathy (HOCM) will be absent. Bedside echocardiography confirms the diagnosis. Why is this important? Approximately 1/3 of patients may go on to develop life-threatening complications if untreated. In particular, ventricular arrhythmias. Close monitoring/follow-up is needed, and permanent ICD should be considered. Management: • Continuous cardiac monitoring, ACS evaluation w/ serial troponin and EKGs • Immediate bedside echocardiography • Cardiology consultation and ultimately ICD placement • Longterm avoidance of competitive sports or other intense physical activities By Dr. Blake Johnson References: 1. Siewe D, Nichols K, Furney S, Littmann L. King of Hearts for Ace of Spades: Apical Hypertrophic Cardiomyopathy. Am J of Med. 2014; vol 127 (1): 31-33. 2. Eriksson MJ, Sonnenberg B, Woo A, et al. Long-term outcome in patients with apical hypertrophic cardiomyopathy. J Am Coll Cardiol. 2002;39(4):638-645. HPI: 55 y/o male with no past medical history presents with vague symptoms including nausea, lethargy, and weakness. Vital signs are within normal limits. Before labs come back you obtain this EKG: EKG: EKG Interpretation: Normal sinus rhythm, rate 67 bpm. Normal axis. Narrow QRS, no ST elevation or depression. --> “Normal ECG”. **On closer inspection you notice the QT interval appears shortened, calculated at 350 ms** Discussion: Initial labs show total calcium of 15.0 mg/dL and PTH of 400 pg/mL (nl 7-53 pg/mL). A single large parathyroid adenoma was diagnosed and subsequently removed. EKG signs of Hypercalcemia: · Short QT interval, usually less than 350 ms · In severe hypercalcemia, can see Osborn waves (J waves) · In extreme hypercalcemia, ventricular irritability and ventricular fibrillation have been reported Remember, degree of EKG findings usually signify severity of hypercalcemia! Administer fluid resuscitation and treat the underlying cause. Simplified DDx for shortened QT interval: 1) Hypercalcemia 2) Congenital short QT syndrome 3) Digitalis toxicity 4) Normal variant. By Dr. Blake Johnson
HPI: Approximately 30 year old female with history of frequent PVCs presents via EMS with palpitations and near syncope. She was driving on the highway today when she had to stop due to abrupt palpitations, SOB, and sensation like she was about to “black out”. EKG: EKG Interpretation:
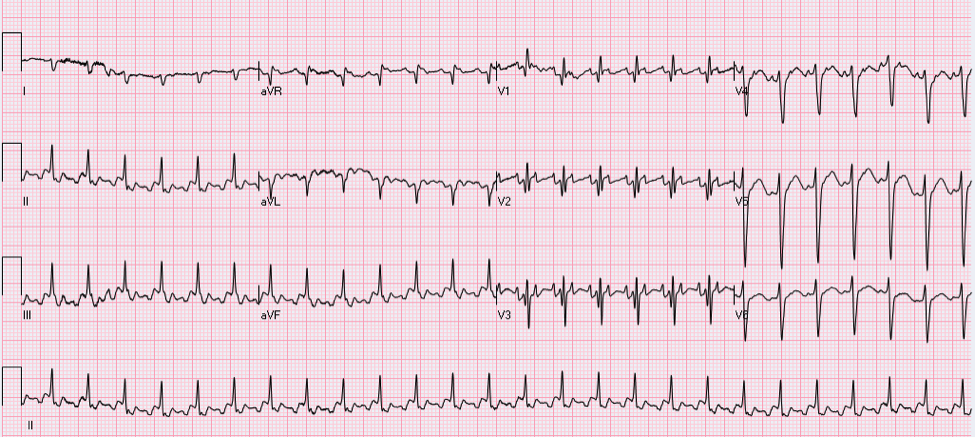
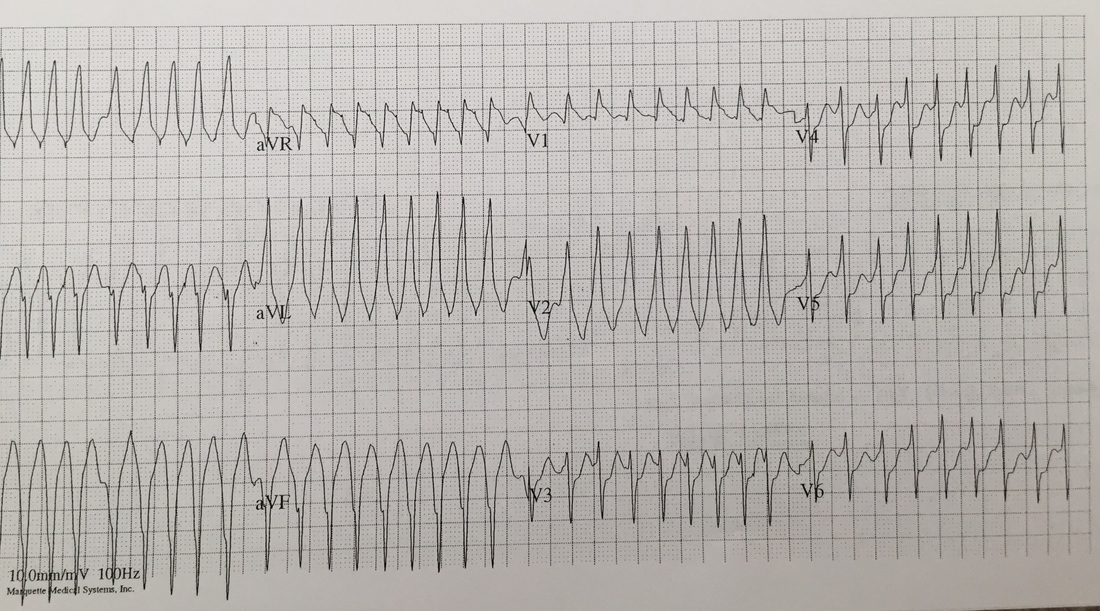
Sinus rhythm with monomorphic PVCs in pattern of bigeminy. Horizontal axis. Inverted T-waves in inferolateral leads (II, III, aVF, V5-V6). Machine says to consider INFEROLATERAL ISCHEMIA. Discussion: Is this ischemia? No. Patient is young and otherwise healthy with no risk factors for cardiovascular disease. She has a history of frequent ventricular ectopy, and presentation is concerning for transient ventricular tachycardia. Pattern of T-wave inversions is consistent with “Cardiac Memory”. How to differentiate: · Clinical scenario not consistent with ACS · Negative T-waves in same leads where PVCs are negative · Eventual resolution after resumption of normal electrical activation Extent and duration of effect correlates with duration of preceding abnormal ventricular conduction. The longer and more profound the electrical disturbance, the longer and more profound Cardiac Memory persists. Most commonly seen with: 1) Ventricular pacing 2) Intermittent LBBB 3) Pre-excitation in WPW 4) Episodes of ventricular ventricular tachycardia (likely cause in our patient). Management: · High index of suspicion for underlying pathology · Provider awareness of phenomenon so as not to initiate unnecessary interventions (Cath lab, etc.) · Treatment aimed at underlying derangement as appropriate · Inpatient vs. outpatient continuous cardiac monitoring if diagnosis in question By Dr. Blake Johnson HPI: Patient is a 54-year-old male presenting with palpitations and near syncope. Patient states that he was told he had a “heart problem” as a child. He was diagnosed with atrial fibrillation some 10+ years ago. EKG: EKG Interperitation:
Rate estimated to be in the high 100s and irregularly irregular. QRS is wide. There are no appreciable P waves. Does not fit any typical bundle branch pattern. Discussion: Is this V tach? No. The patient has a wide complex irregulary irregular waveform. This is MOST likely to be a patient with WPW and a-fib. This is easily confused with ventricular tachycardia. How to differentiate: · Irregularly irregular · Rapid · Wide complex · Does not fit bundle branch pattern · No P waves Treatment: · DO NOT give: adenosine, verapamil, diltiazem, digoxin, beta blocker or amio o Due to the presence of accessory pathway blocking down the AV node, may cause the accessory pathway to become the primary driver of conduction. · Treated with IV procainamide if clinically stable. Dose is 15-16 mg/kg given at a rate no faster than 50 mg/min. · If unstable: Immediate DC cardoversion · Patients will require admission with likely EP study. By Dr Andrew Puchiaty |
EKG ChallengeAuthorER residents on Dr. Littmann's cardiology service present an interesting EKG and core concepts from Dr. Littmann. Archives
January 2016
Categories
All
Disclaimer: All EKG's and images are the sole property of CMC Emergency Medicine Residency and cannot be reproduced without written consent. Patient identifiers have been redacted/changed or patient consent has been obtained. Information contained in this blog is the opinion of the authors and application of material contained in this blog is at the discretion of the practitioner to verify for accuracy.
|

 RSS Feed
RSS Feed
