|
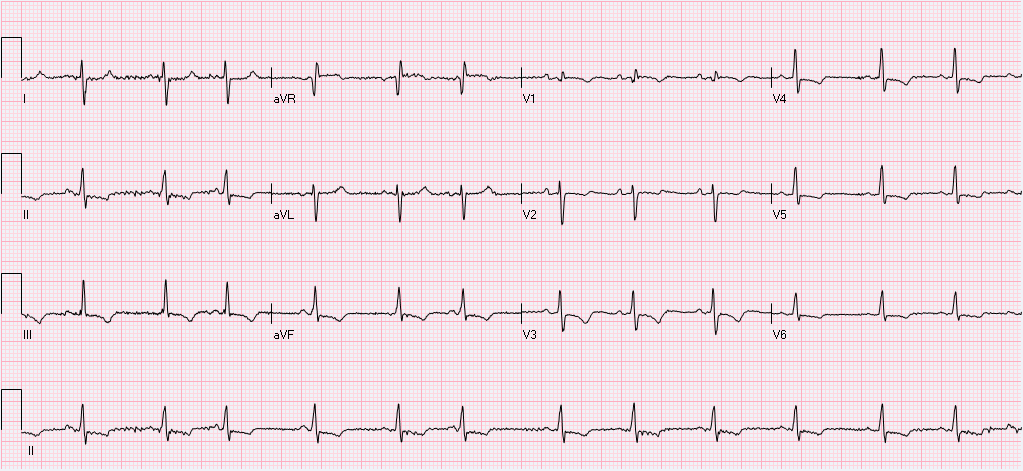
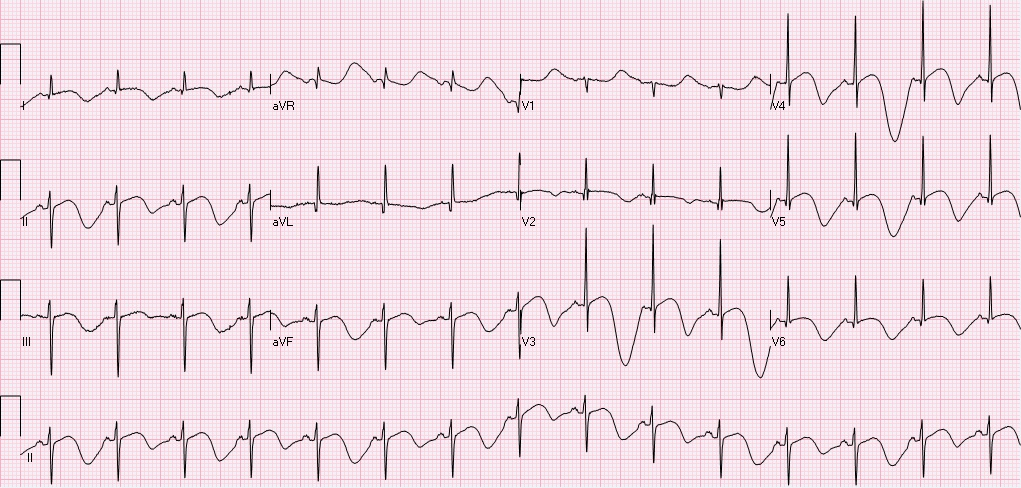
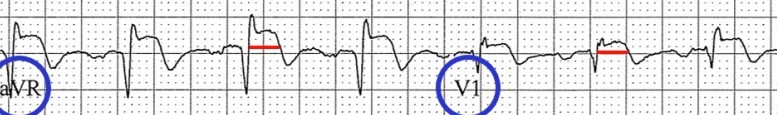
HPI: The patient is a 66 yo F with PMH CHF (TTE 9/2009 with LVEF 50%, RVSP 42), CAD s/p cath ( no stents), DMII, HLD, HTN, and morbid obesity who presents with a cc of abdominal swelling. Onset was 3 months prior to presentation with persistent worsening since that time. She denies any SOB or chest pain/diaphoresis and denies any home O2 use. She does endorse significant weight gain and lower extremity swelling during the same time period. Otherwise, ROS is significant for a fall yesterday, reported to be mechanical without LOC. Labs: remarkable for BNP >2000, no prior value. Troponin 0.01. EKG: Interpretation: Normal sinus rhythm. Rate 76. Right axis deviation with deep S wave in lead I. Normal PR, QRS, and QT intervals. T wave inversions noted in II, III, avF, and all precordial leads. R wave progression which is remarkable for predominantly upgoing QRS in lead V1 with qR complex. As compared to prior EKG(2009), Afib is not present on current. All of the above findings are new.
Conclusion: The patient was subsequently admitted for new onset CHF. Following the echocardiogram which demonstrated the above, the patient was gradually diuresed with plans for RHC to further clarify RVSP with the goal of starting pulmonary vasodilator therapy. RHC was performed 9 days after admission following volume optimization and demonstrated PA 78/36. Pulmonary vasodilator therapy was initiated and the patient was discharged home on these medications. Teaching Points: This patient’s EKG from admission is very consistent with right ventricular hypertrophy and severe pulmonary hypertension. This is evidenced by the new right axis deviation. Further evidence is the qR complex with predominantly upgoing QRS in V1 and T wave inversions inferiorly and in the anterior chest leads. EKG evidence of Pulmonary Hypertension:
Blyth et. al. Quantitative estimation of right ventricular hypertrophy using ECG criteria in patients with pulmonary hypertension: A comparison with cardiac MRI. Pulm Circ. 2011. 470-4. Whitman et al. Validity of the surface electrocardiogram criteria for right ventricular hypertrophy: the MESA-RV study. J Am Coll Cardiol. 2014 Feb 25; 63:672-81. Blog by Dr. Jaron Raper
0 Comments
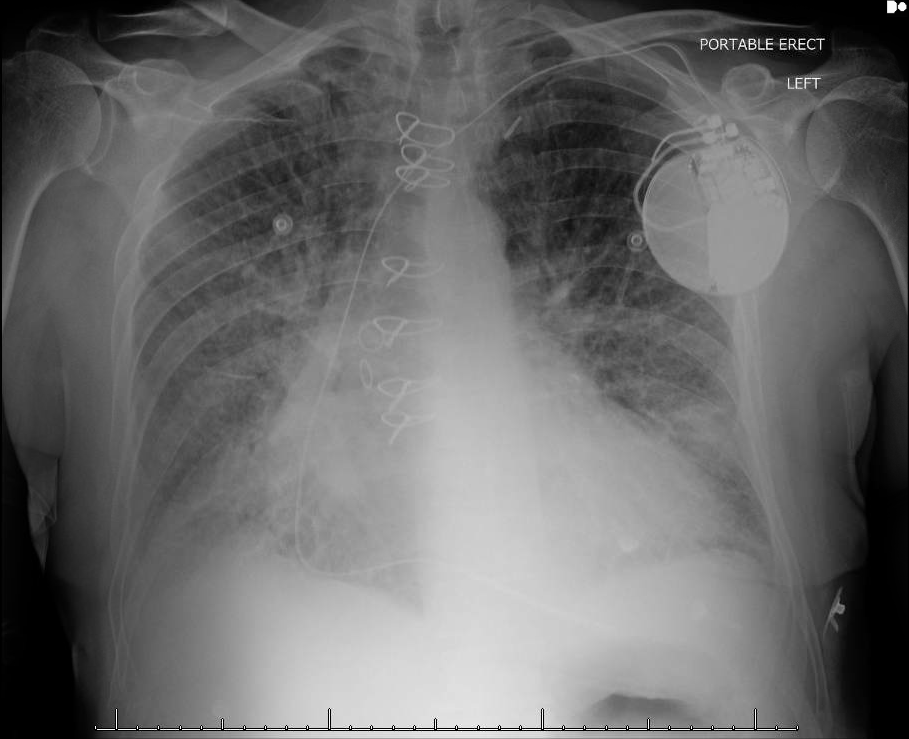
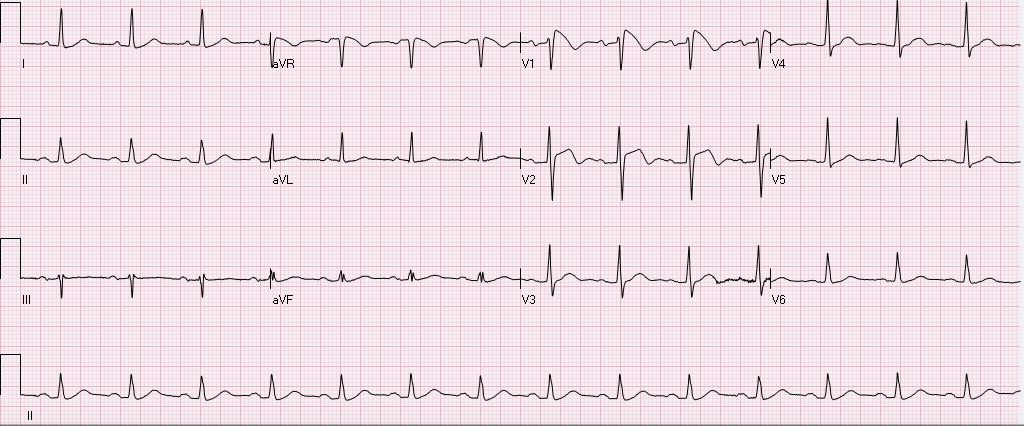
HPI: The patient is a 71 year old male with PMH of ischemic cardiomyopathy (HFrEF 25%) and AICD in place, moderate mitral regurgitation, permanent atrial fibrillation (rate control and fully anticoagulated), HTN, and COPD who presents to the ED with worsening shortness of breath following a fight with his girlfriend a few hours before. He denies any chest discomfort or palpitations but does endorse presyncope with tunnel vision. At onset, he turned his home O2 up to 3L from 1L. Upon EMS arrival, he received solumedrol and a Duoneb treatment without improvement. Upon arrival to the ED the patient was tripoding and in severe respiratory distress. A trial of BiPap in the emergency department was unsuccessful 2/2 continued distress and increased work of breathing, and the patient was subsequently intubated. Exam: significant for tachypnea to 29. Otherwise VS wnl.. Tripoding and accessory muscle use. Irregularly irregular rhythm with blowing holosystolic murmur 3/6 loudest at the left sternal border. CXR: - Interpreted as severe pulmonary edema EKG: EKG (prior to intubation): Atrial fibrillation. Rate 90’s. Normal axis. QRS >140ms. QS in V1 consistent with LBBB. High frequency oscillatory pattern/respiratory artifact noted as highlighted above, consistent with severe respiratory distress. No Sgarbossa criteria are present. Occasional pacer spike noted.
Relevant Laboratory Data: BNP 1400 from prior 1000, Troponin 0.00à0.06, spot cocaine negative Echo: Left atrium and ventricle are dilated. LVEF 25-30% with septal akinesis. Normal valvular function, however prior echo does report 3+ mitral regurgitation. Otherwise unchanged from prior. Interval History: The patient was admitted to MICU, where he was subsequently weaned from the ventilator and extubated within hours of admission following afterload reduction and IV diuresis. Chart review did demonstrate this to be the third admission in the last 2 months for similar symptoms requiring intubation. Prior AICD interrogations demonstrated afib with RVR with a rate of >200, presumed to be the inciting event for respiratory failure. Current management plan (pending) is for repeat AICD interrogation, and nuclear perfusion stress testing to rule out new ischemia. Subsequently the patient will be converted to a BiV pacer with plans for ablation of the His-bundle which should prevent further episodes of RVR and will result in complete pacer dependence. Teaching Points: Respiratory Artifact
Littman L, Rennyson SL, Wall BP, Parker JM. Significance of Respiratory Artifact in the Electrocardiogram. Am J Cardiol. 2008 Oct 15;102(8):1090-6. doi: 10.1016/j.amjcard.2008.05.061. Epub 2008 Jul 25. Blog by Dr. Jaron Raper HPI: The patient is a 69 yo Male with suttering chest pain for the last 8 or 9 days who woke up initially with severe stabbing 10/10 substernal chest pain with concurrent shortness of breath. He did not seek immediate medical attention, but continued to have recurrent episodes of chest pain over the course of the next week which were both exertional and nonexertional. Throughout this same time period he reports fatigue and SOB for which he sought evaluation at his PCP, who referred him to the emergency department. Exam: significant for a systolic ejection murmur 3/6 in intensity heard best over the left sternal border and radiating to the axilla. Radiation is also noted to the carotids bilaterally. The second heart sound is only mildly decreased. Hemoccult positive. Labs: Notable for normocytic anemia with Hgb 7.5. Initial troponin was 0.80 and peaked at 1.14. EKG: Interpretation: Normal Sinus rhythm. Rate 89. Normal axis. Normal PR, QRS, and QT intervals. T wave inversions noted in II, III, and aVF. Poor R wave progression and cannot rule out remote anteroseptal MI evidenced by Q waves in V1,V2, V3.
Cath: The patient was subsequently cath’d and found to have 100% mid-distal RCA occlusion which is consistent with inferior ischemia previously noted on EKG. LAD ostial 50% and mid 50% stenosis. Echo: significant for mild aortic stenosis with mean gradient 22 mmHg. Conclusion: Following catheterization, no stenting was performed and plans for follow-up with GI for colonoscopy to determine source of occult bleed. Cath did confirm a remote RCA infarct presumed to be exacerbated by the patient’s anemia. Aortic stenosis was not considered severe enough to generate Heyde’s syndrome (mean gradient of <40mmHg) Teaching Points: Heyde's Syndrome
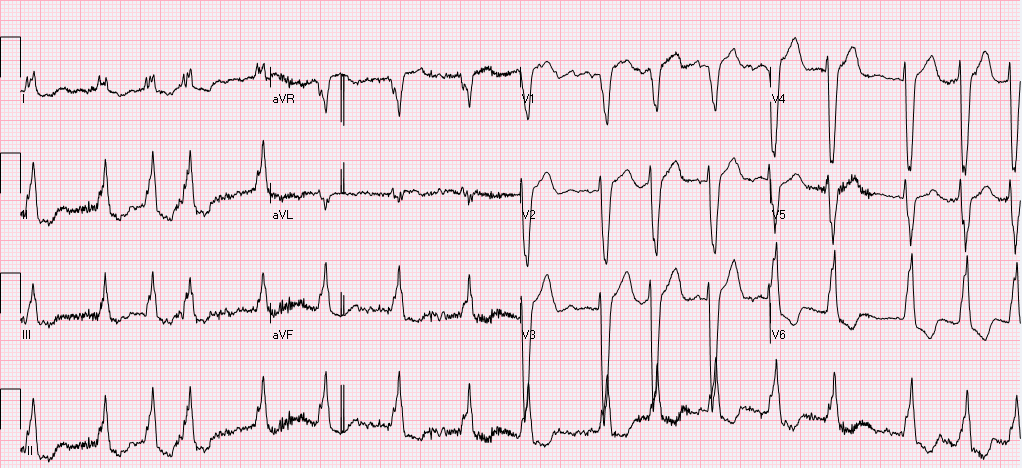
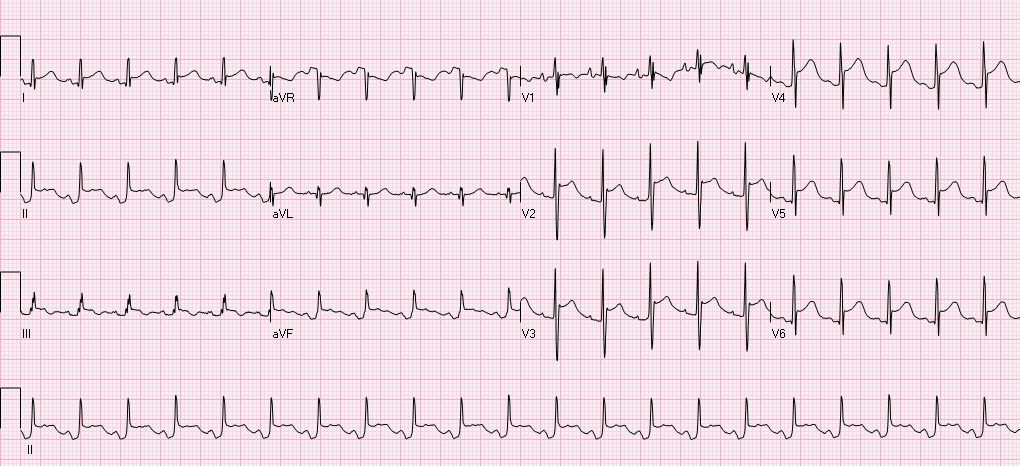
Vincentelli A, Susen S, Le Tourneau T, Six I, Fabre O, Juthier F, Bauters A, Decoene C, Goudemand J, Prat A, Jude B. Acquired von Willebrand syndrome in aortic stenosis. N Engl J Med. 2003;349(4):343. Blog by Dr. Jaron Raper A 56 year old female with a history of asthma, hypothyroidism, tobacco use, chronic opioid use was found down at home, found to have subarachnoid hemorrhage, left temporal hematoma with bilateral supraclinoid aneurysms on CTA, s/p embolization. Hospitalization has been complicated by vasospasm, fevers, long QT, and stress induced cardiomyopathy. On hospital day 11 the following EKG was obtained to assess the patient's long QT noted on previous EKGs (500, 516, 620). Rate 89, sinus rhythm with sinus p waves, left axis deviation, normal PR, QRS intervals, prolonged QTc 732. T-wave inversions in I, II, III, aVF, V2-V6. In fact, if you pay attention to the T-wave morphology you will notice polymorphic deep t-waves, alternating in depth and morphology. There are two interesting points from this EKG: 1) T-QT pattern, and 2) T wave alternans. T-QT pattern:
T wave alternans:
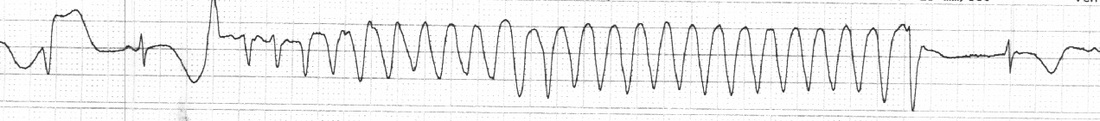
Approximately 2 hours after the above EKG, the following rhythm strip was noted on telemetry. Here you see a PVC followed by a compensatory pause, followed by polymorphic VT. Pauses are frequent triggers of Torsades. The patient went on to experience several sustained, and non-sustained runs of polymorphic VT overnight. She received aggressive Mg & K repletion, defibrillator pads were placed, but patient remained hemodynamically stable and did not require defibrillation or overdrive pacing. QT decreased and VT resolved.
Teaching points:
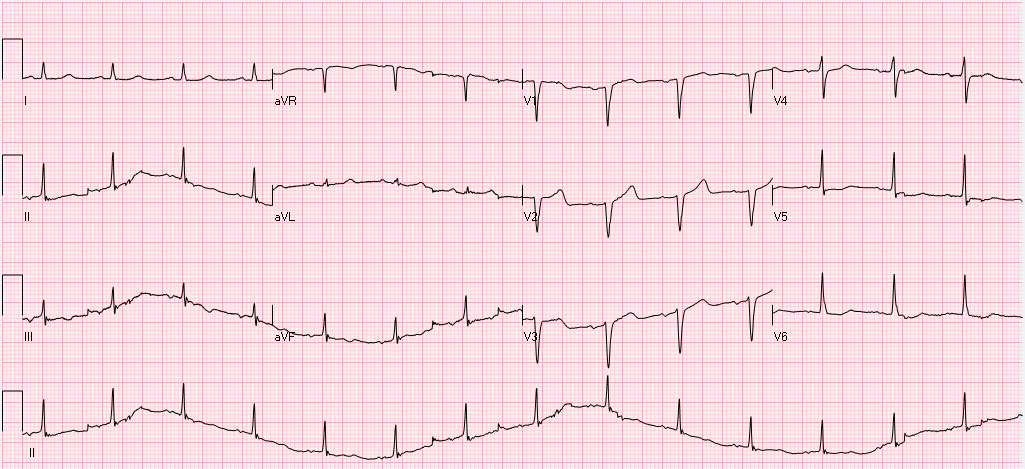
References: Warraich H, Buxton A, Kociol R. Macroscopic T-wave alternans in a patient with takotsubo cardiomyopathy and QT prolongations. Heart Rhythm. 2014 Oct;11(10):1848-9. Sommargren C, Drew B. Preventing torsades de pointes by careful cardiac monitoring in hospital settings. AACN Adv Crit Care. 2007 Jul-Sep;18(3):285-93 Clinical context: This is a 20s year-old male who presented to the ED after GSW to the thorax. He lost pulses in transport, and underwent ED thoracotomy, cardiac massage and aortic cross clamping with ROSC, and subsequent ex-lap. He has been in the ICU with complications including respiratory failure, renal failure, hemorrhagic shock, and multiple returns to the operating room for intrabdominal injuries. The following EKG was obtained over the weekend due to ST elevation noted on cardiac telemetry. Automated read notes sinus tachycardia, ****ACUTE MI/STEMI****
Let's systematically review this ECG. The ventricular rate is 126, there is a p wave before every QRS - sinus rhythm, QRS is upgoing in I, II - normal axis, PR, QRS intervals appropriate, QTC prolonged at 492, there is ST elevation in I, II, III, aVF, V2-V6 with concomitant PR depressions, reciprocal ST depressions and PR elevations in aVR and V1. This is a classic ECG for PERICARDITIS. The patient subsequently had cardiac ultrasound that demonstrated a small effusion as well. Remember that classic findings of pericarditis include widespread ST elevation and PR depression with reciprocal changes in aVR +/- V1. Life in the fast lane review of pericarditis While there are many causes of pericarditis, this patient has several underlying risk factors including chest trauma, surgery, and possible infection secondary to limited sterility in the emergency setting. While the reported incidence of acute pericarditis after general thoracic surgery is very low, I was not able to identify any recent literature addressing the incidence of pericarditis after ED thoracotomy in patients without penetrating cardiac injuries. In patients with penetrating cardiac injury delayed secondary cardiac injuries and complications occur at at rate of ~50%. Colchicine is noted to be used as pericarditis prophylaxis in major cardiac surgery and transplant, this could be considered in the ED setting, however the logistics and lack of evidence as to how common this complication is could limit benefit. Complications of ED Thoracotomy include:
Upon return to the OR the day after thoracotomy this patient was noted to have ongoing bleeding from internal mammary arteries, but no damage to coronary arteries or other cardiac damage. References: Imazio, M., Gaita, F., LeWinter, M. Evaluation and Treatment of Pericarditis A Systematic Review. JAMA. 2015;314(14):1498-1506. Fallahnejad M., Kutty AC., Wallace HW. Secondary lesion of penetrating cardiac injuries: a frequent complication. Ann Surg. 1980;191(2):228. Alraies MC, AL Jaroudi W, Shabrang C, et al. Clinical features associated with adverse events in patients with post-pericardiotomy syndrome following cardiac surgery. Am J Cardiol. 2014 Nov;114(9): 142630. Keller D, Kulp H, Maher Z, et al. Life after near death: Long term outcomes of Emergency department thoracotomy survivors. J Trauma Acue Care Surg. 2013 May;74(5):1315-20. HPI: A previously healthy, incarcerated, 43 year old Asian American male presented to the ED after a syncopal episode with subsequent VF arrest. The patient is successfully defibrillated. After ROSC the following EKG is obtained: Normal rate, regular rhythm, sinus P waves in from of QRS. normal PR, normal QRS width, axis, amplitude, progression. ST elevation in V1, V2, inverted T waves in V2, normal QTc.
After obtaining this EKG the patient was rushed to the cath lab, no obstructive coronary lesions were identified. What is the diagnosis, and what is the treatment? Is this finding always pathological? The diagnosis is Brugada syndrome. - syndrome is a constellation of Brugada pattern EKG + CLINICAL SYMPTOMS Brugada Pattern EKG: -RBBB pattern in V1 (rSR', terminal positive QRS complex) -high take off, upward convex, ST elevation in V1 and V2 -inverted T waves in V1 and V2 Clinical Symptoms: syncope, +/- palpitations, unexplained VF/arrest, family history of sudden death. Brugada Syndrome: -due to poorly understood sodium chanellopathy. There is NO UNDERLYING STRUCTURAL HEART DISEASE. -except for V1-V2, the EKG is usually normal. -Brugada pattern on EKG can be transient, and can be unmasked by physical stressors, medications, metabolic derangements, and ischemia. Treatment: ICD is indicated for patients with Brugada syndrome. Brugada Pattern EKG is NOT ALWAYS pathalogical. -Brugada pattern EKG can be a normal variant, especially in a patient without risk factors, symptoms, and the pattern is an incidental finding during a non-cardiac workup. -Brugada pattern can also be seen in severe hyperkalemia, sodium-channel blocker toxicity, propofol infusion syndrome, right ventricular pathology or injury. Patients who are incidentally found to have Brugada pattern EKG without cardiac symptoms are at low risk for sudden cardiac death. These patients should be educated on finding, referred to cardiologist, and given careful follow up precautions if they develop syncope, palpitations, chest pain, or other cardiac symptoms. A careful family history to elucidate family members with sudden cardiac death is warranted. There is some risk if the patient has Brugada EKG pattern without symptoms, but does have a family member who experienced sudden cardiac death. These patients should be referred to a cardiologist, and may undergo provocative testing to induce VT/VF, although this is controversial. References: 1. Littmann, L. Monroe, MH., Kerns, WP., Svenson, RH., Gallagher, JJ. Brugada syndrome and "Brugada sign": clinical spectrum with a guide for the clinician. Am Heart J. 2003 May;145(5):768-78. 2. http://lifeinthefastlane.com/ecg-library/brugada-syndrome/ HPI: 83 year old caucasian male with a past medical history of atrial fibrillation, COPD, HTN, metastatic prostate cancer, presented to ED after a ground level fall, found to have altered mental status, cerebral edema with midline shift, no intracranial bleeding. EKG: Auto read: Demand pacemaker; interpretation is based on intrinsic rhythm. Atrial fibrillation with premature ventricular or aberrantly conducted complexes. Cannot rule out Inferior infarct, age undetermined. ST & T wave abnormality, consider anterolateral ischemia or digitalis effect. Abnormal ECG. No previous ECGs available.
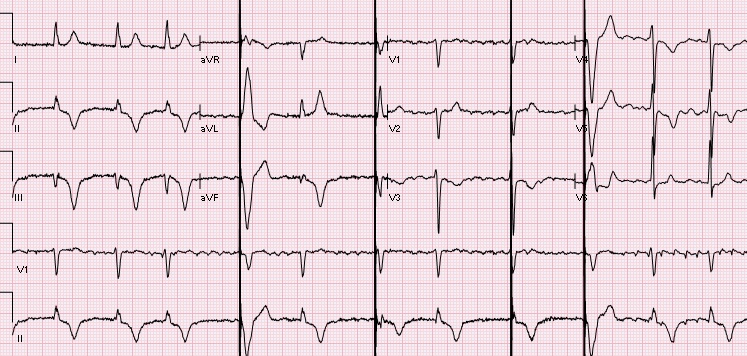
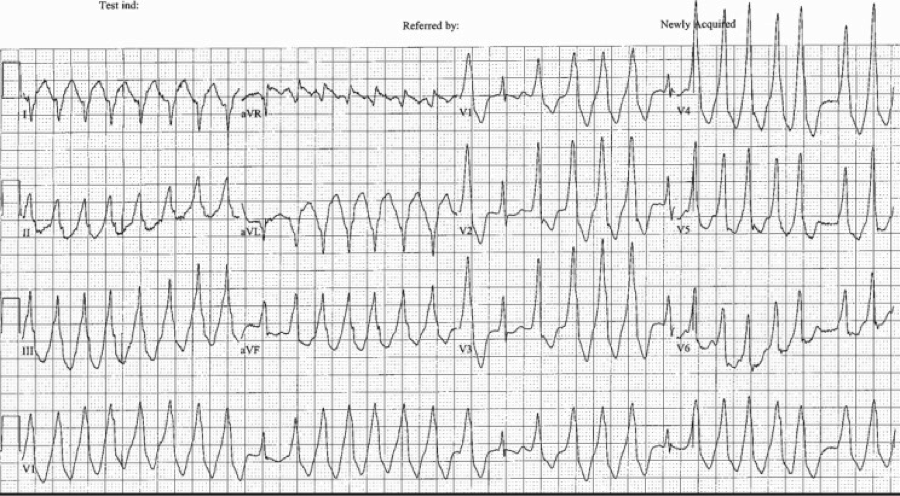
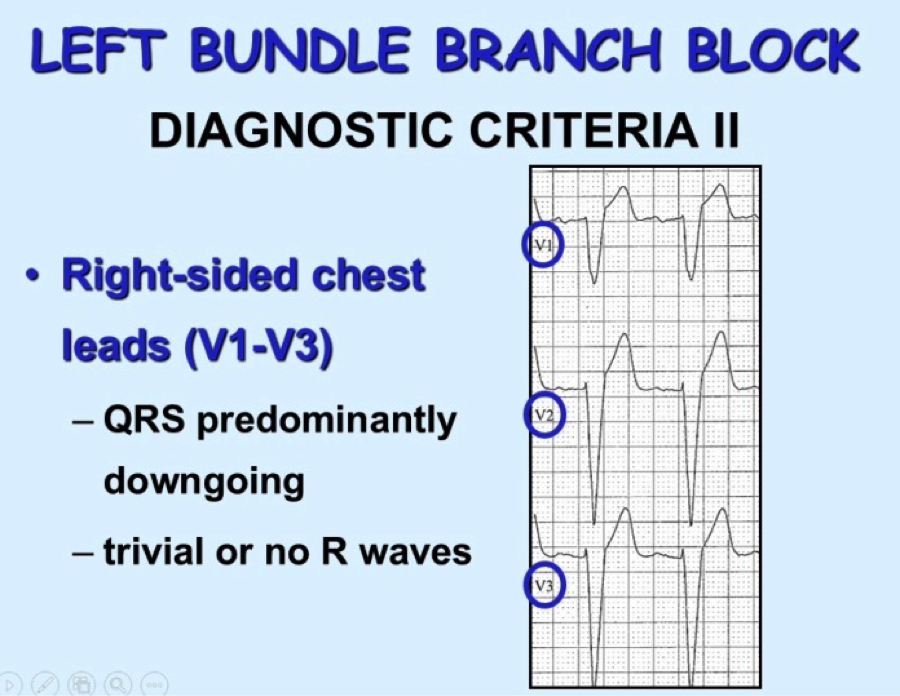
Let's discuss this EKG. Ventricular rate is in the 60-70s, rhythm is definitely not sinus, as there are not a p wave before every QRS. The P waves, in fact, are absent. Axis is normal (upgoing QRS in I & aVF). The QRS is upgoing in Lead I suggestive of left ventricular pacing. QRS is not wide, QT is not prolonged. There are deep negative T waves in II, III, aVF, V4, and V5. There is normal R wave progression. Deep T waves in this EKG are a result of a phenomenon known as Cardiac Memory. These are NON-ISCHEMIC T wave inversions that are a result of repolarization abnormalities in the setting of intermittently wide QRS. So what causes these non-ischemic t-wave inversions? Well, first the QRS must become intermittently wide, as is seen in demand pacing (such as in the patient in this case), WPW, prolonged VT, or intermittent LBBB. Then, as the QRS becomes narrow again, the T waves become negative in the leads where QRS was wide. There is a lag in resolution of the repolarization abnormality, which is likely channel related (but poorly understood). The persistence of T wave inversion depends on how frequently the QRS is wide, or in this case, how frequently the heart is LV paced. Basically the T waves are slow to normalize after an episode of widened QRS. There is a broad differential diagnosis for T wave inversion, including subendocardial ischemia, acute CNS bleeds, acute adrenergic stress, PE, metabolic abnormalities, and drug effects. Because of the broad differential, new diffuse T wave inversion should always focus on the "worst first", and in general cardiac memory is a diagnosis of exclusion. Recognizing this characteristic post-pacing T-wave morphology can be a valuable piece of information in your clinical decision making. Ischemia workup is not indicated based on the EKG alone. This transient EKG finding, in conjunction with negative serum cardiac enzymes, absence of chest pain/cardiac complaints, makes cardiac memory the most likely cause of the EKG abnormality. Don't fear the deep inverted T waves in demand pacing! References: Littman, L., Fermant, A. Large T-Wave Inversion in a Patient With a Pacemaker. Arch Intern Med. 2011 Aug 8;171(15):1314-16. HPI: Teenage female with SOB, presyncope, and palpitations x 1 hour. HR is 220 in triage. BP 118/84, RR 28, SpO2 96%. Alert and oriented. EKG: Question: What arrhythmia is this? How you would treat this rhythm in a stable patient. What if the patient were unstable? ECG Interpretation: Rate of 173/min. Irregularly irregular rhythm, wide complex tachycardia with intermittent narrow complexes and changing of QRS morphologies. No LBBB or RBBB pattern. Diagnosis: Afib with WPW Let’s discuss the interpretation of irregularly irregular Wide Complex Tachycardias (WCT), and then we will discuss the treatment. At first glance, it is difficult to assess the regularity, but with some scrutiny, you can see that the rhythm is irregular. The differential diagnosis of irregularly irregular WCT is essentially limited to three main dysrhythmias. Dr. Littmann calls these FBI: Fast, Broad, and Irregular:
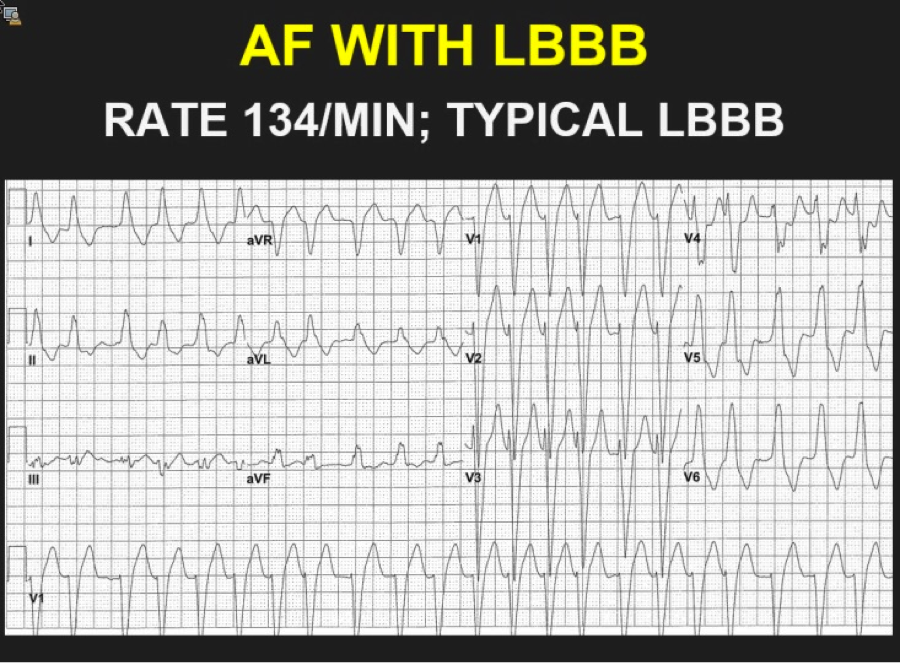
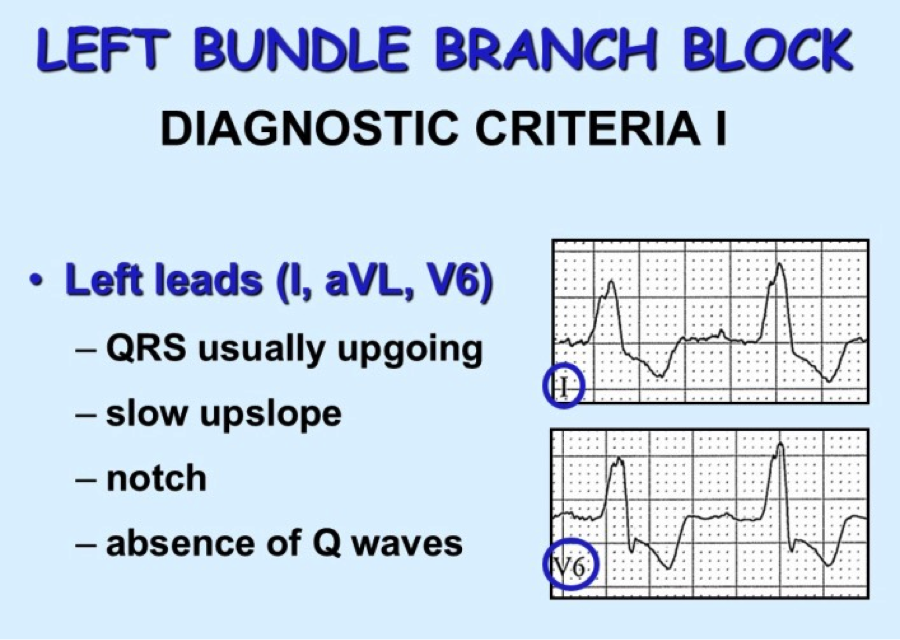
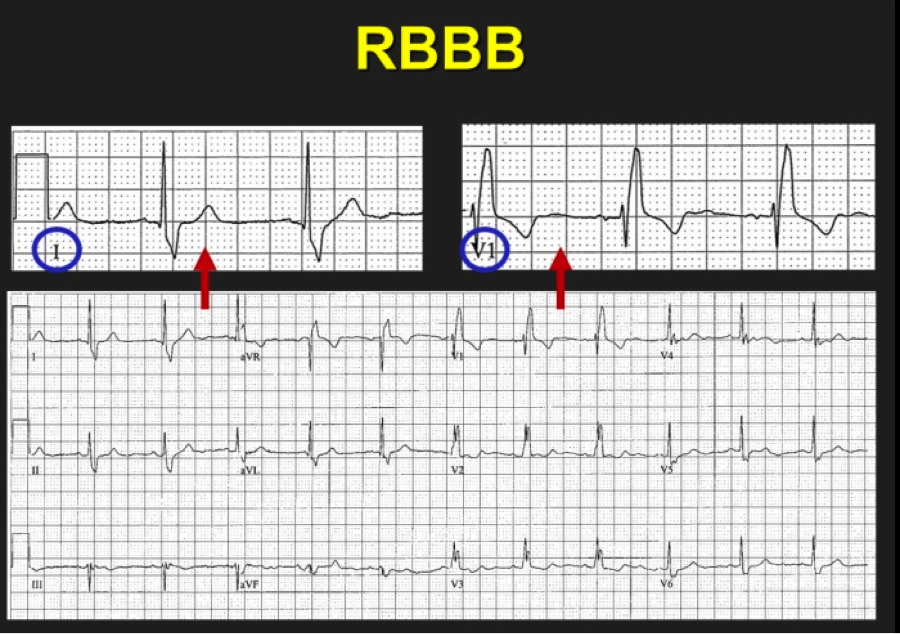
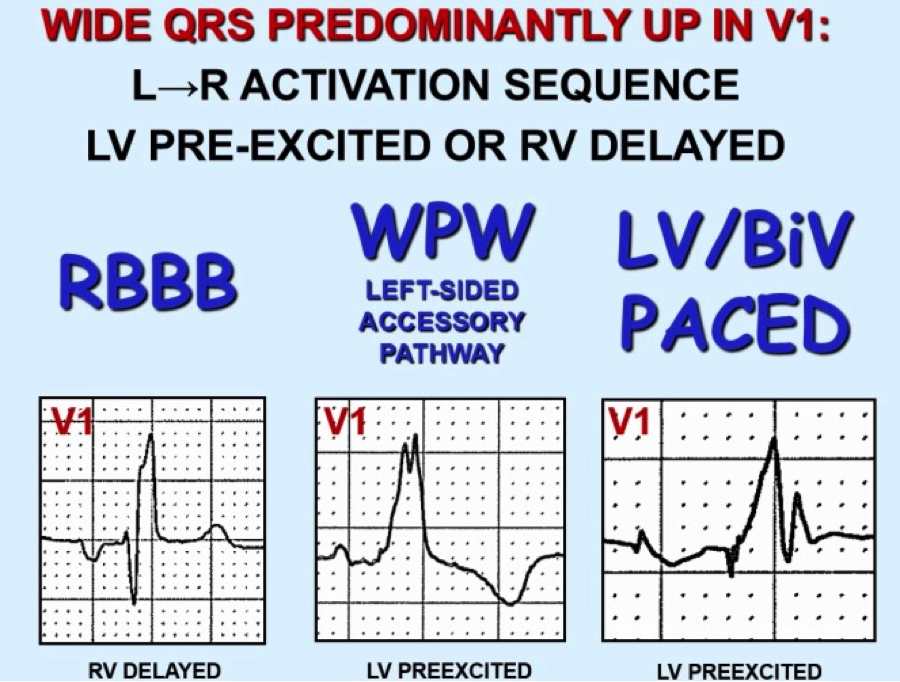
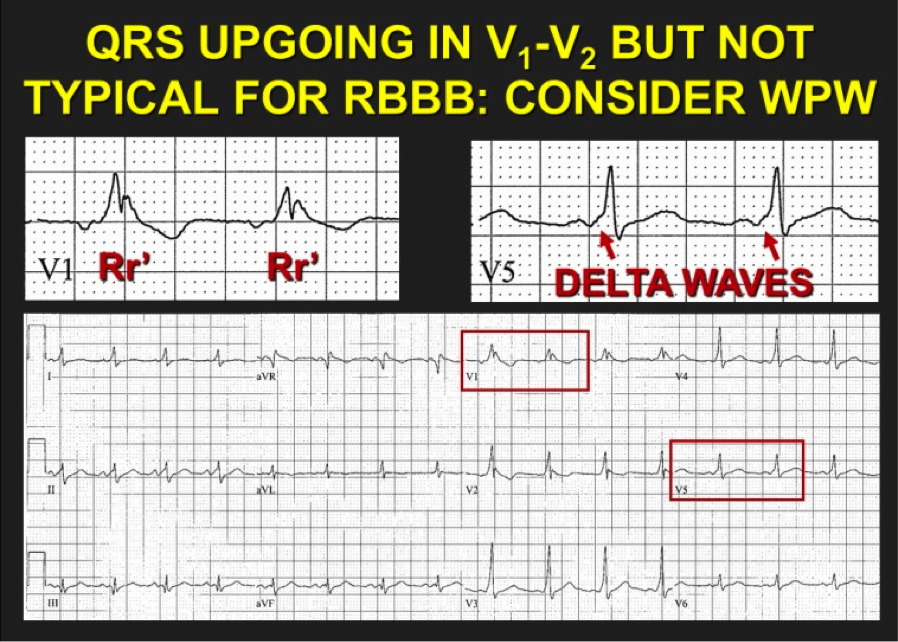
The above ECG is an irregularly irregular WCT meeting the criteria for LBBB as below (upgoing in I, deep S waves in V1). Note the rate of 134. Fast, but not extremely fast, indicating likely transmission through the AV node. Consider RBBB, looking for triphasic QRS, with rSR’ and 2nd upgoing phase wider and taller than first upgoing phase as shown in these images: So we have scrutinized our irregularly irregular WCT and found no evidence of BBB pattern. We are now left with Afib with WPW. Why is this important? The treatment is completely different from regular WCT (usually Vtach) and Afib with BBB pattern. Treatment of FBI, Fast Broad and Irregular:
Key Points:
As a reminder for recognizing WPW in the non-tachycardic patient’s ECG: References:
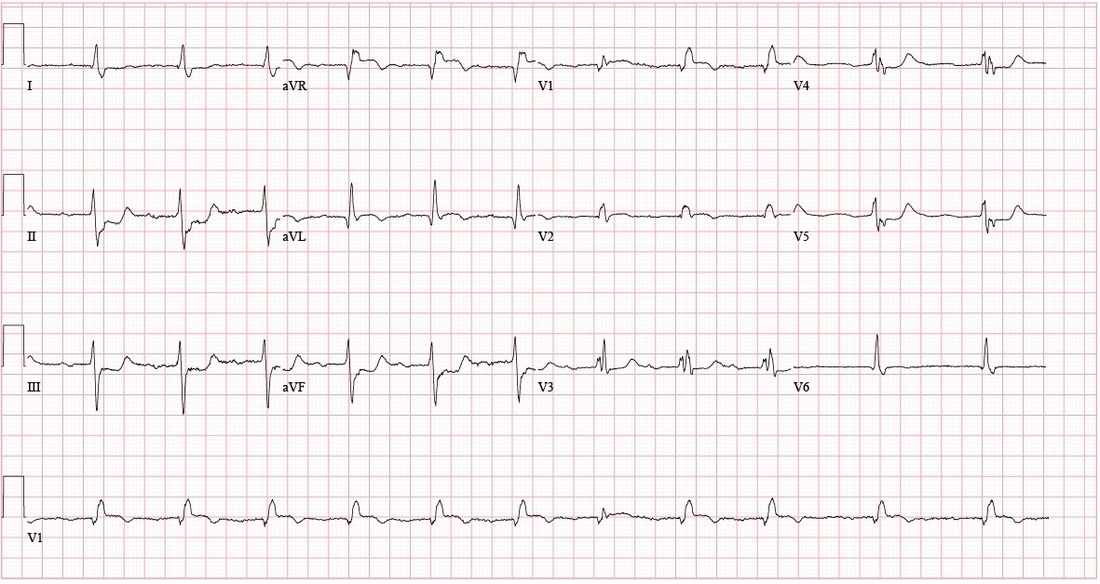
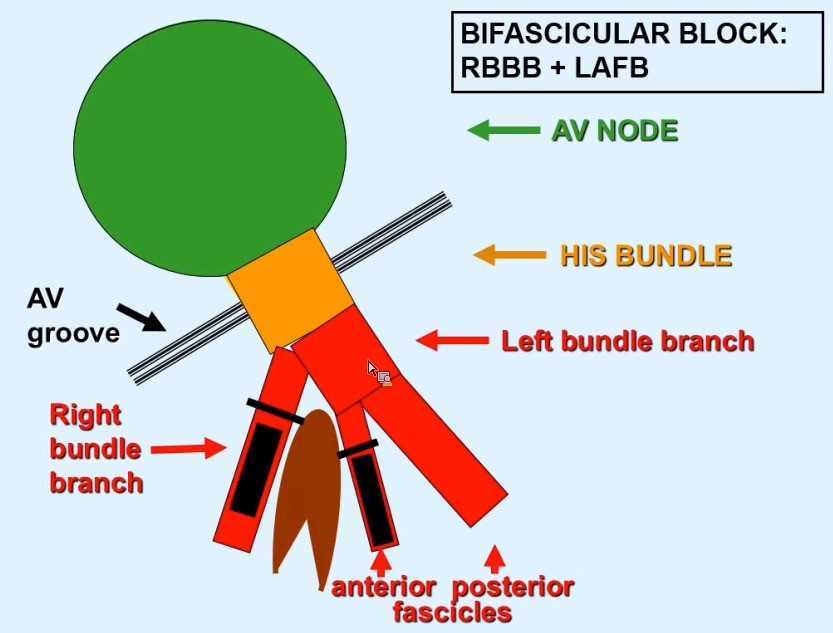
By: Dean Tanner, PGY1 EM HPI: 90 yo male with PMH of HTN, DM presents to ED with 2-3 weeks of lower extremity edema and intermittent chest pain, with acute worsening of cardiac chest pain yesterday and this morning, now accompanied by SOB, nausea, and lightheadedness. Evaluated in major and found to have a heart rate 40s-60s. ECG: Questions: How is a bifascicular block diagnosed? What are the clinical implications of the bifascicular block? What about in the context of a first degree AV block? ECG Interpretation: Sinus rhythm, rate 69, with sinus arrhythmia and First Degree AV Block. PR interval 272. QRS 150. QTc 490. Left Axis Deviation. (QRS is upward in I and downward in II) Right BBB + Left Anterior Fascicular Block = Bifascicular Block Subtle ST Elevation V1 and V2; consider anterolateral STEMI ST depression inferior leads; inferior ischemia vs. reciprocal changes from anterolateral STEMI Discussion: Definitions: (LAFB = Left Anterior Fasicular Block; RAFB = Right Anterior Fascicular Block )
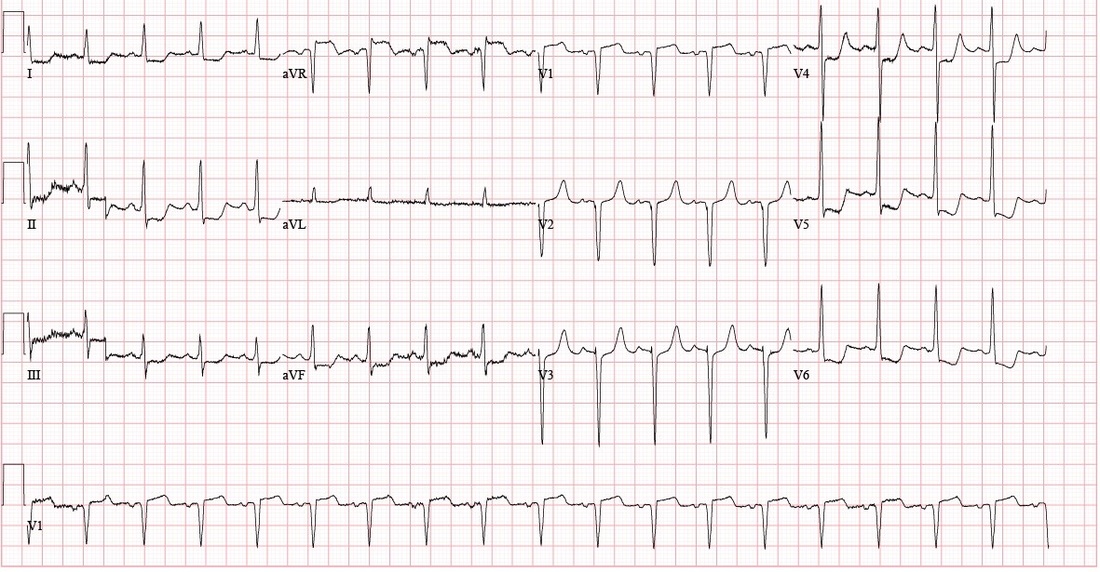
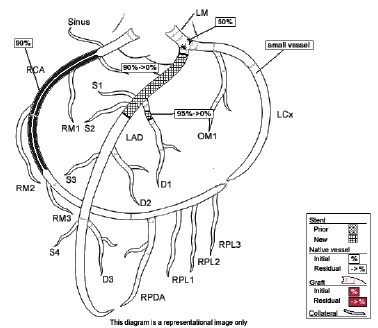
• Bifascicular block: RBBB + either LAFB or RAFB o RBBB + Left Axis Deviation (<-45) = LAFB o RBBB + Right Axis Deviation (>+90)= RAFB • Incomplete Trifascicular block: Conduction damage to all three fascicles o Bifascicular block + first degree AV block OR o Bifascicular block + second degree AV Block OR o RBBB+ Alternating LAFB/LPFB Implications: o In an asymptomatic patient, bifascicular block is largely incidental and no workup is indicated. o If the patient presents with syncope and bifascicular block, this is a medical emergency and the American College of Cardiology and AHA recommend pacemaker implantation. o Risk of progression to complete heart block thought to be low (1% per year in a cohort of 554 patients), but some studies have suggested rates as high as 16% in RBBB + LAFB and 21-75% in RBBB + RAFB. Higher risk in a retrospective review prior to modern reperfusion therapy. o Patients with incomplete trifascicular block are thought to have a greater risk of progression to complete heart block, though data are not conclusive Patient course: Admitted to ICU with systolic pressure in 70s, felt to be cardiogenic shock secondary to MI. Patient was treated with 1 amp of atropine, which improved heart rate from 40s to 70s. Initial troponin was 3.41, which plateaued at 84 on 9/8. No ST elevation. Poor cath candidate given age, AKI at presentation. Treated medically (ASA, statin, Plavix, heparin gtt, carvedilol) and has been transferred to the floor, now hemodynamically stable. Last ECG relatively unchanged since admission (persistent first degree AV block, RBBB, LAFB, ST depression. References: • Dr. Littmann’s ECG Lectures (graphic also from Dr. Littmann) • http://lifeinthefastlane.com/ecg-library/basics/bifascicular-block/ • Uptodate sections on “Conduction Abnormalities after Myocardial Infarction” and “Course and Treatment of Bifascicular Block” • ACC/AHA/HRS 2008 Guidelines for Device-Based Therapy of Cardiac Rhythm Abnormalities: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Writing Committee to Revise the ACC/AHA/NASPE 2002 Guideline Update for Implantation of Cardiac Pacemakers and Antiarrhythmia Devices): developed in collaboration with the American Association for Thoracic Surgery and Society of Thoracic Surgeons. Circulation. 2008;117(21):e350. By: Dean Tanner, PGY1 HPI: 50-year-old man with PMH of HTN, DM, ESRD on dialysis, and polysubstance abuse with a one-year history of recurrent chest pain and multiple visits to the emergency department for chest pain exacerbations. He was recently seen for left-sided chest pain with an EKG showing nonspecific ST depressions, negative chest x-ray, and elevated troponins ×3, peaked at 1.31. He did not undergo cardiac catheterization due to recent cocaine use. He returns today with 10/10 chest pain refractory to nitro taken at home. ECG: ECG Interpretation: Sinus tachycardia with rate of 108. PR and QRS intervals normal. Probable LVH. Anterior Q waves, possibly due to LVH. ST depression inferiorly and laterally. ST elevation 2 mm in aVR and 1 mm in V1. Patient course: Underwent successful emergent cardiac catheterization. LAD 90-95% occlusion, LAD-1st Diagonal 90% occlusion, LMCA 50% occlusion, proximal RCA 90% occlusion (stented later). Peak troponin 26.70. Discussion: aVR sign: possible Left Main Coronary Artery (LMCA) occlusion or other severe coronary disease (proximal LMA, 3-vessel disease) • When associated with angina and diffuse ST-segment depression o ST elevation in aVR ≥ 1mm is 80% specific for severe LMCA disease or 3-vessel disease. (LAD + LAD-D1 in this case) o ST elevation in aVR > V1 typically signifies LMCA over proximal LAD o If STE in aVR is not > V1, more likely proximal LAD occlusion
o STE in aVR is associated with very high mortality. STE > 1mm associated with 6-7 fold increase in mortality STE > 1.5mm associated with 20-75% mortality • When associated with STE of other leads, aVR STE is independently associated with increased mortality. Pro Tips: • Patients with ≥ 1 mm STE in aVR may potentially require early/urgent CABG; discuss with cardiology or CT surgery prior to giving Clopidogrel/Prasugrel (thienopyridines 7 or less days before CABG is associated with increase in major bleeding). • Presence of aVR sign may not meet criteria for a Code STEMI, but it should be recognized as a STEMI equivalent and and requires urgent reperfusion therapy References: • http://lifeinthefastlane.com/ecg-library/lmca/ • Kosuge M, Ebina T, Hibi K, Morita S, Endo M, Maejima N, Iwahashi N, Okada K, Ishikawa T, Umemura S, et al. An early and simple predictor of severe left main and/or three-vessel disease in patients with non-ST-segment elevation acute coronary syndrome. Am J Cardiol. 2011;107:495–500 • Tamura, A. Significance of lead aVR in acute coronary syndrome. World Journal of Cardiology, 2014; 6(7): 630-637. • Dr. Littmann’s ECG video lectures |
EKG ChallengeAuthorER residents on Dr. Littmann's cardiology service present an interesting EKG and core concepts from Dr. Littmann. Archives
January 2016
Categories
All
Disclaimer: All EKG's and images are the sole property of CMC Emergency Medicine Residency and cannot be reproduced without written consent. Patient identifiers have been redacted/changed or patient consent has been obtained. Information contained in this blog is the opinion of the authors and application of material contained in this blog is at the discretion of the practitioner to verify for accuracy.
|


 RSS Feed
RSS Feed
