|
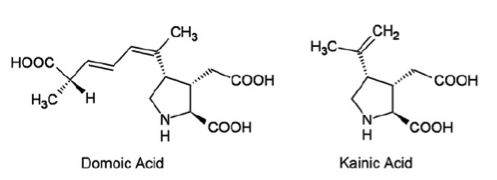
Introduction: Domoic acid is a marine excitatory neurotoxin secreted by pseudo-nitzschia diatoms and various species of chondria armata red algae which are found throughout the world. The toxin is bio-accumulated in marine animals such as shellfish, anchovies and sardines and is then passed to humans who ingest them. It is temperature stable when accumulated in the tissues of shellfish and is not inactivated by cooking or freezing. The toxin is a water soluble, tri-carboxylic acid which is very closely related to kainic acid, a neurotoxin found in seaweed and is a structural analogue of endogenous glutamic and aspartic acids. Domoic acid induces a constellation of symptoms in humans known as Amnesic Shellfish Poisoning. Only one recorded event of this has occurred and took place in 1987 on the eastern coast of Prince Edward Island. 147 patients ingested mussels containing the toxin. 1/3 of these eventually developed neurologic symptoms including permanent short term memory deficits, seizure and coma. There were 4 fatalities. As of 2015 the western coast of the United States is currently experienced the largest pseudo-nitzschia bloom in ten years prompting closure of many commercial fisheries. Structure: Pharmacology/Pharmacokinetics: Domoic acid is a water soluble tri-carboxylic acid that is absorbed orally. Systematic investigations of volume of distribution do not appear to have been yet undertaken. However, given its chemical structure as a hydrophilic organic acid its lipophilicity is likely low. Similarly it should have difficultly crossing the blood brain barrier. Studies have shown that the molecule is excreted almost exclusively by the kidneys with the molecule largely intact suggesting very little in vivo metabolism. Studies conducted in rats showed a renal clearance rate of less than 10 ml/min/kg. This appears to show large variability between species with primate studies showing an excretion approximately ten times slower than that observed in rats.
Toxic doses also appear to vary greatly between species. Data obtained in humans is scarce and is largely restricted to data gathered during the 1987 Prince Edward Island event. Findings shown below: Oral Dose (mg/kg) Clinical Symptoms - 0.2-0.3 No observable effects - 0.9- 2.0 GI symptoms - 1.9-4.2 Seizure, disorientation, coma Interestingly, Domoic Acid has also been used as an anti-helminthitic in Japan for quite some time. One study from the late 1950’s reports doses of 0.4-0.8 mg/kg given to Japanese children with no apparent side effects. The LD50 in studies conducted in mice was found to be 3.6 mg/kg. Domoic acid does appear to concentrate in the amniotic fluid of pregnant mammals with one study showing 24% of the serum value. It is not readily cleared from this space and thus likely poses significant developmental risks. It does appear to enter breast milk of lactating mammals as well. Metabolic Pathways and active metabolites: Available data does not show evidence of extensive metabolism in mammals. It appears to be excreted largely intact almost exclusively through the kidneys. Toxicity/Mechanism of Toxicity: Domoic acid is an excitatory neurotoxin and appears to exert effects on ionotropic glutamatergic receptors in the CNS, particularly the AMPA, NMDA and kainite subtypes. It binds with high affinity to these receptors causing an excessive influx of calcium which disrupts intracellular calcium currents, triggering apoptosis and leading to neuronal death. The mechanism is very similar to glutamatergic excitatory toxicity. Of note, the hippocampus contains a high concentration of susceptible receptors and is thus disproportionately affected likely leading to the signature clinical finding of permanent short term memory deficits. Clinical Toxicity/Presentation: The clinical presentation during acute exposure is characterized by two phases of symptoms. Mild acute GI symptoms such as nausea and vomiting are often seen within the first 24 hours. CNS symptoms such as headache, dizziness, confusion, disorientation, seizures, short term memory loss, motor weakness, and coma are generally seen in the first 48 hours following exposure. Other side effects observed included cardiac dysrhythmia, hypotension, and increased respiratory secretions. Studies performed in sea lions which are primary consumers of the pseudo-nitzschia diatom and thus chronically exposed have shown that chronic sub-lethal exposure may cause epilepsy and cardiomyopathy. The demographic appeared to be most heavily affected were those over 65 years of age. This is likely due to decreased renal function and decreased integrity of the blood-brain barrier secondary to aging. Laboratories: Clinical lab studies are likely of little use in the acute phase beyond a chemistry screen to assess renal function. Rapid ELISA as well as chromatography are available for the detection of domoic acid directly. Treatment/Management: Largely supportive. Data from the 1987 incident showed that seizures were largely able to be managed by benzodiazepines and phenytoin. Due to the renal excretion of the toxin, dialysis may theoretically play a role in those with renal impairment, however this has not yet been investigated. By Dr. Andrew Johnson, PGY2 References 1. Teitelbaum, MD, J., Zatorre, Ph.D, R., Carpenter, MD, S., Gendron, MD, D., Evans, Ph.D, A., Gjedde, MD, A., & Cashman, MD, N. (1990). Neurologic Sequelae of Domoic Acid Intoxication Due to the Ingestion of Contaminated Mussels. New England Journal of Medicine, 322(25), 1781-1787. 2. Pulido, O. (2008). Domoic Acid Toxicologic Pathology: A Review. Marine Drugs, 6(2), 180-219. 3. Jeffery, B., & Barlow, T. (2004). Amnesic Shellfish Poisoning. Food Chemical Toxicology, 42(4), 545-557. 4. Ramsdell, J., & Frances, G. (2014). Domoic Acid Epileptic Disease. Marine Drugs, 12(3), 1185-1207. 5. Lefebvre, K., & Robertson, A. (2010). Domoic acid and human exposure risks: A review. Toxicon, 56, 218-230.
1 Comment
Wolfman ?
9/8/2019 05:33:22
What happens when domoic acid is mixed with fog outside and people are in it
Reply
Your comment will be posted after it is approved.
Leave a Reply. |
Toxicology BlogAuthorEM Rotators on Toxicology Selected by Feedspot as one of the Top 20 Toxicology Blogs on the web
Archives
March 2018
Categories
All
Disclaimer: All images included on this blog are the sole property of CMC EM Residency and cannot be used or reproduced without written permission. Patient identifiers have been redacted/changed or patient consent has been obtained. Information contained in this blog is the opinion of the author and application of material contained in this blog is at the discretion of the practitioner to verify for accuracy.
|

 RSS Feed
RSS Feed
