|
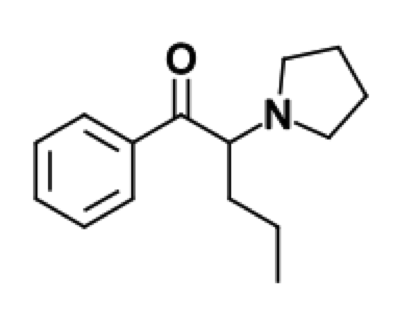
36 year old male with PMHx of IDDM and depression was prescribed a new drug by his psychiatrist for psychogenic erectile dysfunction. He presented to the hospital 16 hours after taking 350mg of this drug. He was drowsy and confused on presentation with a BP of 130/80 and HR of 150 with atrial fibrillation, which he had no history of. He was having rigors and retrosternal pain. He had no other co-ingestions. His rectal temperature was 35.5C. He was well-perfused peripherally. His laboratory studies were otherwise unremarkable. He was admitted for altered mental status and for new atrial fibrillation, which spontaneously resolved within 24 hours.
Pharmacology/Uses:
Use in erectile dysfunction:
Pharmacokinetics:
Clinical toxicity:
Treatment:
References
Other sources used include Goldfrank’s 10th edition, Haddad’s 3rd edition, and The Poison Review (www.thepoisonreview.com) Authored by: Dr. Michael Mollo M.D.
1 Comment
20% intravenous fat emulsion (IFE) may be useful to resuscitate severe cardiotoxicty due to fat-soluble drugs; the so-called lipid rescue. Propofol is a fat soluble anesthetic agent that is formulated and delivered in 3% IFE. If 20% IFE is not immediately available, can I use propofol as a substitute for 20% IFE? What is Intravenous Lipid Emulsion?
How does it work?
What can it treat?
How do you give it?
Are there any adverse reactions?
What lab tests are altered?
Why do people think Propofol will work?
Have any studies been performed?
* Propofol in intralipid group had longer time to each adverse outcome than the other groups
What did I take away from this?
References:
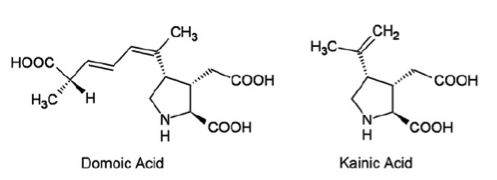
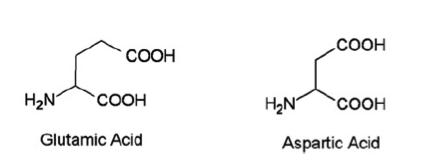
Introduction: Domoic acid is a marine excitatory neurotoxin secreted by pseudo-nitzschia diatoms and various species of chondria armata red algae which are found throughout the world. The toxin is bio-accumulated in marine animals such as shellfish, anchovies and sardines and is then passed to humans who ingest them. It is temperature stable when accumulated in the tissues of shellfish and is not inactivated by cooking or freezing. The toxin is a water soluble, tri-carboxylic acid which is very closely related to kainic acid, a neurotoxin found in seaweed and is a structural analogue of endogenous glutamic and aspartic acids. Domoic acid induces a constellation of symptoms in humans known as Amnesic Shellfish Poisoning. Only one recorded event of this has occurred and took place in 1987 on the eastern coast of Prince Edward Island. 147 patients ingested mussels containing the toxin. 1/3 of these eventually developed neurologic symptoms including permanent short term memory deficits, seizure and coma. There were 4 fatalities. As of 2015 the western coast of the United States is currently experienced the largest pseudo-nitzschia bloom in ten years prompting closure of many commercial fisheries. Structure: Pharmacology/Pharmacokinetics: Domoic acid is a water soluble tri-carboxylic acid that is absorbed orally. Systematic investigations of volume of distribution do not appear to have been yet undertaken. However, given its chemical structure as a hydrophilic organic acid its lipophilicity is likely low. Similarly it should have difficultly crossing the blood brain barrier. Studies have shown that the molecule is excreted almost exclusively by the kidneys with the molecule largely intact suggesting very little in vivo metabolism. Studies conducted in rats showed a renal clearance rate of less than 10 ml/min/kg. This appears to show large variability between species with primate studies showing an excretion approximately ten times slower than that observed in rats.
Toxic doses also appear to vary greatly between species. Data obtained in humans is scarce and is largely restricted to data gathered during the 1987 Prince Edward Island event. Findings shown below: Oral Dose (mg/kg) Clinical Symptoms - 0.2-0.3 No observable effects - 0.9- 2.0 GI symptoms - 1.9-4.2 Seizure, disorientation, coma Interestingly, Domoic Acid has also been used as an anti-helminthitic in Japan for quite some time. One study from the late 1950’s reports doses of 0.4-0.8 mg/kg given to Japanese children with no apparent side effects. The LD50 in studies conducted in mice was found to be 3.6 mg/kg. Domoic acid does appear to concentrate in the amniotic fluid of pregnant mammals with one study showing 24% of the serum value. It is not readily cleared from this space and thus likely poses significant developmental risks. It does appear to enter breast milk of lactating mammals as well. Metabolic Pathways and active metabolites: Available data does not show evidence of extensive metabolism in mammals. It appears to be excreted largely intact almost exclusively through the kidneys. Toxicity/Mechanism of Toxicity: Domoic acid is an excitatory neurotoxin and appears to exert effects on ionotropic glutamatergic receptors in the CNS, particularly the AMPA, NMDA and kainite subtypes. It binds with high affinity to these receptors causing an excessive influx of calcium which disrupts intracellular calcium currents, triggering apoptosis and leading to neuronal death. The mechanism is very similar to glutamatergic excitatory toxicity. Of note, the hippocampus contains a high concentration of susceptible receptors and is thus disproportionately affected likely leading to the signature clinical finding of permanent short term memory deficits. Clinical Toxicity/Presentation: The clinical presentation during acute exposure is characterized by two phases of symptoms. Mild acute GI symptoms such as nausea and vomiting are often seen within the first 24 hours. CNS symptoms such as headache, dizziness, confusion, disorientation, seizures, short term memory loss, motor weakness, and coma are generally seen in the first 48 hours following exposure. Other side effects observed included cardiac dysrhythmia, hypotension, and increased respiratory secretions. Studies performed in sea lions which are primary consumers of the pseudo-nitzschia diatom and thus chronically exposed have shown that chronic sub-lethal exposure may cause epilepsy and cardiomyopathy. The demographic appeared to be most heavily affected were those over 65 years of age. This is likely due to decreased renal function and decreased integrity of the blood-brain barrier secondary to aging. Laboratories: Clinical lab studies are likely of little use in the acute phase beyond a chemistry screen to assess renal function. Rapid ELISA as well as chromatography are available for the detection of domoic acid directly. Treatment/Management: Largely supportive. Data from the 1987 incident showed that seizures were largely able to be managed by benzodiazepines and phenytoin. Due to the renal excretion of the toxin, dialysis may theoretically play a role in those with renal impairment, however this has not yet been investigated. By Dr. Andrew Johnson, PGY2 References 1. Teitelbaum, MD, J., Zatorre, Ph.D, R., Carpenter, MD, S., Gendron, MD, D., Evans, Ph.D, A., Gjedde, MD, A., & Cashman, MD, N. (1990). Neurologic Sequelae of Domoic Acid Intoxication Due to the Ingestion of Contaminated Mussels. New England Journal of Medicine, 322(25), 1781-1787. 2. Pulido, O. (2008). Domoic Acid Toxicologic Pathology: A Review. Marine Drugs, 6(2), 180-219. 3. Jeffery, B., & Barlow, T. (2004). Amnesic Shellfish Poisoning. Food Chemical Toxicology, 42(4), 545-557. 4. Ramsdell, J., & Frances, G. (2014). Domoic Acid Epileptic Disease. Marine Drugs, 12(3), 1185-1207. 5. Lefebvre, K., & Robertson, A. (2010). Domoic acid and human exposure risks: A review. Toxicon, 56, 218-230. Introduction:
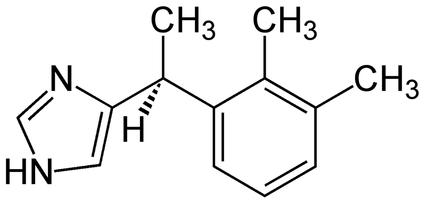
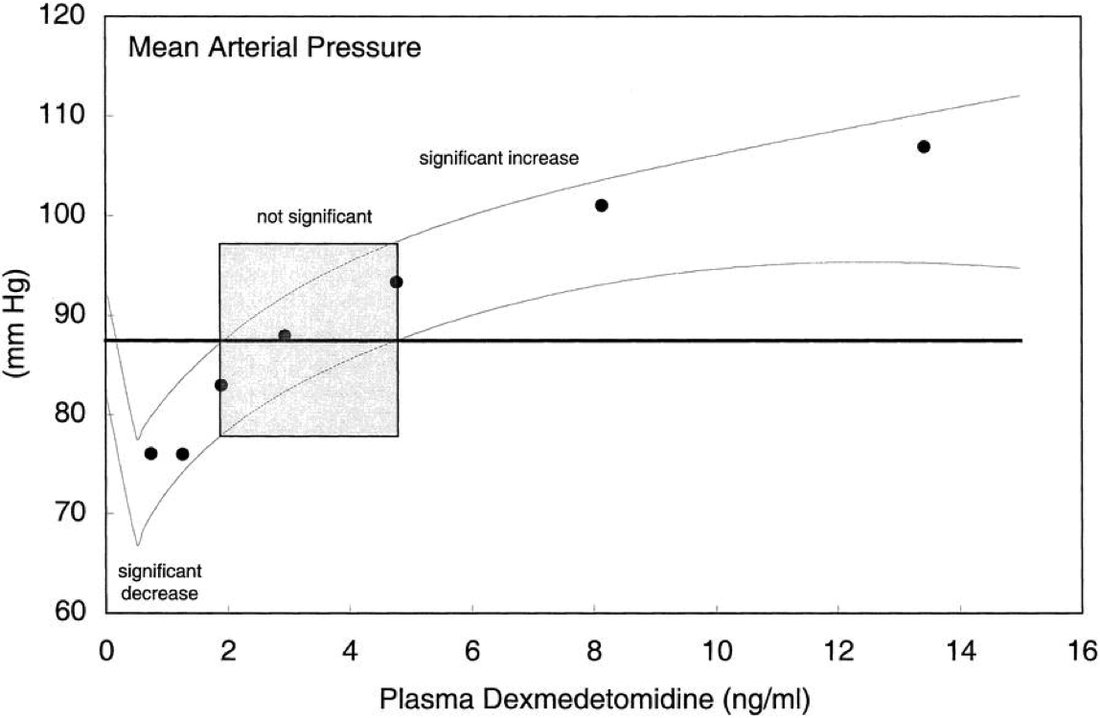
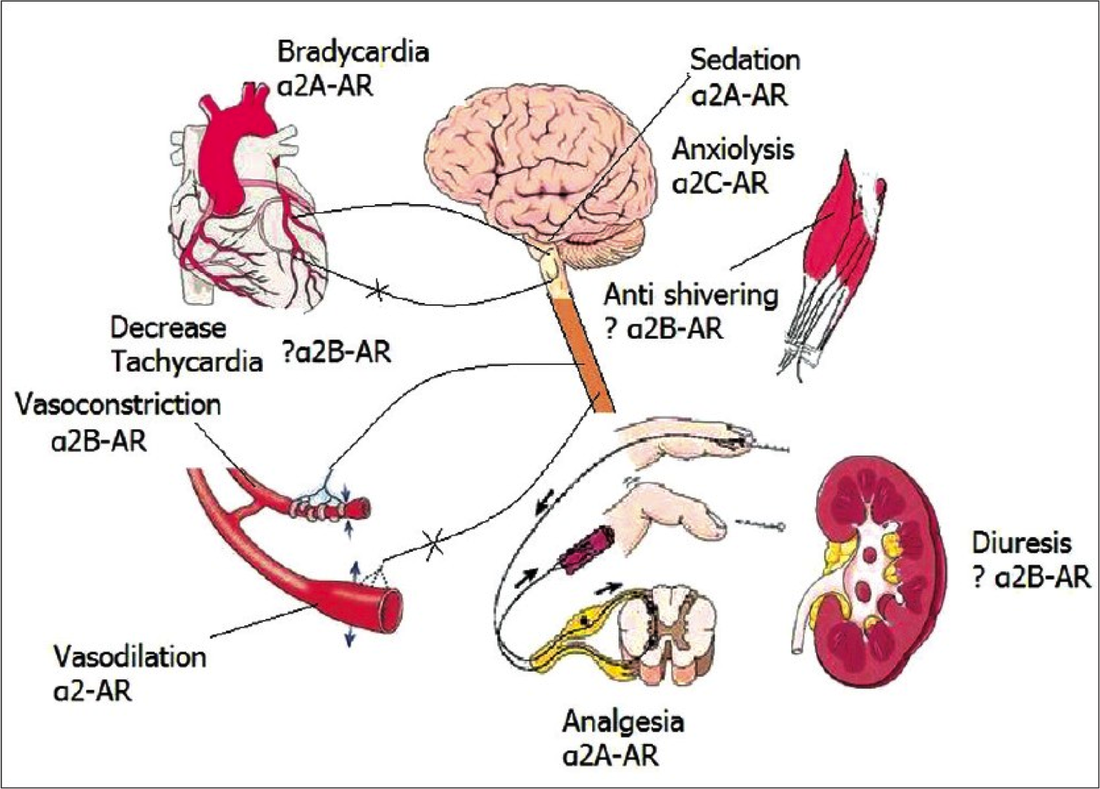
Structure: C13H17CIN2 Pharmacology/Pharmacokinetics: Comparing Alpha2 Agonists
Dosing:
Absorption/Distribution/Metabolism/Excretion:
Significant Drug/Drug Interactions:
Toxicity/Mechanism of Toxicity:
Treatment/Management:
By Dr. Patrick Jackson, MD References: ∗ Albertson T, Chenoweth J, Ford J, et. al. Is it prime time for alpha2-adrenocepter agonists in the treatment of withdrawal syndromes? J Med Toxicol. 2014; 10(4):369-381. ∗ Bryczkowski S, Lopreiato M, Yonclas P, et. al. Risk factors for delirium in older trauma patients admitted to the surgical intensive care unit. J Trauma Acute Care Surg. 2014; 77(6):944-951. ∗ Crispo A, Daley M, Pepin J, Harford P, Brown C. Comparison of clinical outcome in nonintubated patients with severe alcohol withdrawal syndrome treated with continuous-infusion sedatives: Dexmedetomidine versus benzodiazepines. Pharmacotherapy. 2014; 34(9):910-917. ∗ DeMuro J, Botros D, Wirkoski E, Hanna A. Use of dexmedetomidine for the treatment of alcohol withdrawal syndrome in critically ill patients: A retrospective case series. J Anesth. 2012; 26(4)601-605. ∗ Flower O, Hellings S. Sedation in traumatic brain injury. Emerg Med Int. 2012; e-published. ∗ Lizotte R, Kappes J, Barte B, et. al. Evaluating the effects of dexmedetomidine compared to propofol as adjunctive therapy in patients with alcohol withdrawal. Clin Pharmacol. 2014; 31(6):171-177. ∗ Lonardo N, Mone M, Nirula R, et. al. Propofol is associated with favorable outcomes compared with benzodiazepines in ventilated intensive care unit patients. American Journal of Respiratory and Critical Care Medicine. 2014; 189(11):1383-1394. ∗ Mancl E, Brophy G. Time to wake up: A historical perspective on modernized sedation management. Society of Critical Care Medicine. 2013; e-published. ∗ Mazanikov M, Udd M, Kylanpaa L, et. al. Dexmedetomidine impairs success of patient-controlled sedation in alcoholics during ERCP: A randomized, double blind, placebo-controlled study. Surg Endosc. 2013; 27(6):2163-2168. ∗ Muller S, Preslaski C, Kiser T, et. al. A randomized, double-blind, placebo-controlled dose range study of dexmedetomidine as adjunctive therapy for alcohol withdrawal. Crit Care Med. 2014; 42(5):1131-1139. ∗ Muzyk A, Kerns S, Brudney S, Gagliardi J. Dexmedetomidine for the treatment of alcohol withdrawal syndrome: Rationale and current status of research. CNS Drugs. 2013; 27(11):913-920. ∗ Rayner S, Weinert C, Jepsen S, Broccard A. Dexmedetomidine as adjunct treatment for severe alcohol withdrawal in the ICU. Ann Intensive Care. 2012; 23(1):12. ∗ Riihioja P, Jaatinen P, Haapalinna A, et. al. Effects of dexmedetomidine on rat locus coeruleus and ethanol withdrawal symptoms during intermittent ethanol exposure. Alcohol Clin Exp Res. 1999; 23(3):432-438. ∗ Riihioja P, Jaatinen P, Oksanen H, et. al. Dexmedetomidine, diazepam, and propranolol in the treatment of ethanol withdrawal symptoms in the rat. Alcohol Clin Exp Res. 1997; 21:801-804. ∗ Roberts D, Hall R, Kramer A, et. al. Sedation for critically ill adults with severe traumatic brain injury: A systematic review of randomized controlled trails. Crit Care Med. 2011; 39(12)2743-2751. ∗ Tolonen J, Rossinen J, Alho H, Harjola V. Dexmedetomidine in addition to benzodiazepine-based sedation in patients with alcohol withdrawal delirium. Eur J Emerg Med. 2013; 20(6):425-427. ∗ Traeger J, Popa A, Makii J. Dexmedetomidine for acute alcohol withdrawal. Society of Critical Care Medicine. 2014, e-published. ∗ Savel R, Kupfer Y. Using dexmedetomidine as adjunctive therapy for patients with severe alcohol withdrawal syndrome: Another piece of the puzzle. Crit Care Med. 2014; 42(5)1298-1299. ∗ VanderWeide L, Foster C, MacLaren R, et. al. Evaluation of early dexmedetomidine addition to the standard of care for severe alcohol withdrawal in the ICU: A retrospective controlled cohort study. J Intensive Care Med. 2014; e-published. ∗ Wong A, Benedict N, Kane S. Multicenter evaluation of pharmacologic management and outcomes associated with severe resistant alcohol withdrawal. J Crit Care. 2015; 30(2):405-409. Alpha-PVP (aka “Flakka” or “Gravel”) Introduction: -Also referred to as Alpha-pyrrolidinovalerophenone, alpha-pyrrolidinopentiophenone, (RS)-1-phenyl-2-(1-pyrrolidinyl)-1-pentanone -Cathinone is a natural stimulant found in the khat plant -Synthetic cathinones, aka “Bath salts”, refer to a collective group of compounds that are highly potent stimulants and are similar to MDMA, cocaine, and methamphetamine -The most common compound found in bath salts is MDPV (3,4-methylenedioxypyrovalerone) -Alpha-PVP is not a “second generation bath salt”; it was actually first synthesized in the 1960’s -Alpha-PVP has been recently been linked to multiple reported deaths in Florida -Alpha-PVP was added to the controlled substance list in the USA in 2014 Structure: -Alpha-PVP typically comes in a crystal form -Formula: C15H21NO Pharmacology/Pharmacokinetics:
-Alpha-PVP’s mechanism of action is believed to involve inhibition of the reuptake of norepinephrine, dopamine, and serotonin -Rat studies have showed that alpha-PVP is both an uptake blocker of norephinephrine/dopamine and stimulates dopamine release Absorption/Distribution/Metabolism/Excretion: Absorption -Can be taken orally, sublingually, injected, vaporized, or intranasal -Onset of desired effects (euphoria) in 5-30 minutes and lasts for 1-3 hours. However, the undesired side effects (including psychosis) can last days Distribution -Postmortem studies have found alpha-PVP uniformly distributed among multiple tissues (blood, brain, muscle, CSF, lung, kidney, and liver) Metabolism -Six phase I metabolites have been identified -Lactam is the major metabolite Excretion -A study of postmortem alpha-PVP levels showed the highest concentrations were found in urine and the lowest concentrations were found in the liver → suggesting the drug is excreted chiefly by the renal system Significant Drug/Drug Interactions: -Co-ingestion of alcohol, other stimulants, and other illicit drugs has been shown to enhance the effects of the drugs -A large proportion of the fatal cases of alpha-PVP have involved co-ingestions of multiples drugs and alcohol Toxicity/Mechanism of Toxicity: -LD50 has not been determined -The potency and efficacy of alpha-PVP and MDPV are very similar in rat studies -Rat studies showed increased locomotor signs with doses ranging from 1mg/kg to 30mg/kg -A few case reports of drug-related fatalities with serum alpha-PVP levels have been reported: 0.1mg/L, 0.29mg/L, 0.52mg/L, 0.901mg/L -All serum levels determined by liquid-mass-mass spectrometry method -There is a significant overlap between concentrations tolerated by individuals and those reported in drug-related fatalities → alpha-PVP concentration alone does not determine toxicity Clinical Toxicity/Presentation: -Increased dopamine causes euphoria, increased activity, and hyperstimulation -Increased norepinephrine increases heart rate and blood pressure -Increased serotonin can cause hallucinations, delirium, and paranoia -Severe agitated delirium and aggressive behavior have been reported -Life threatening severe toxicity: Serotonin syndrome, hyperthermia, hypotension, rhabdomyolysis, acute renal failure, liver failure, cardiac dysrhythmias, seizures -Chronic exposure: May lead to physical and psychological dependence with withdrawal symptoms Laboratories/Where it is made: -Mainly manufactured in China and India and then shipped to America by legal delivery companies -Unknown if manufactured in America currently Treatment/Management: -Treatment is symptomatic and supportive -Benzodiazepines for sedation/delirium/seizures/tachycardia/hypertension -IVF’s for dehydration frequently indicated -GI decontamination is not recommended. Single dose of activated charcoal may be considered for possible co-ingestion, but usually not clinically indicated. Hemodialysis is not effective for elimination. -Labs: Obtain electrolytes, renal function, hepatic enzymes, and CPK level -Serum drug levels are NOT clinically useful and NOT readily available -Obtain EKG and continuous cardiac monitoring -Frequent temperature monitoring and institute cooling measures as warranted (Hyperthermia is an indicator of severe toxicity) -Dispo: Minimal observation of 6-8 hours. If patient has persistent CNS stimulation, persistent tachycardia, seizures, or dysrhythmias →then warrants admission -Poison center should be consulted By: Dr. Patrick Jackson References:
Propofol basics:
Mechanism PRIS:
Clinical features:
Risk factors:
Prevention:
Treatment:
References: Diedrick, D, Brown D. Propofol Infusion Syndrome in the ICU. Journal of Intensive Care Medicine. 2011; 26 (2) 59-72. . Rosen DJ, Nicoara A, Koshy N, Wedderburn RV. Too much of a good thing? Tracing the history of the propofol infusion syndrome. J Trauma. 2007;63(2):443-447.
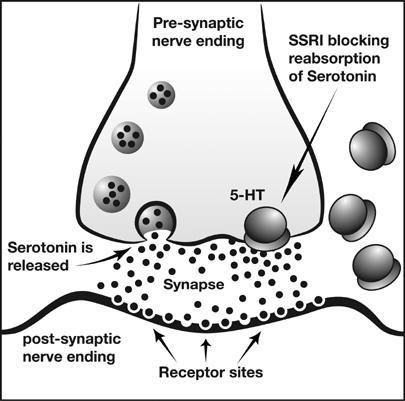
 Bleeding in Pregnancy: SSRI use 1-2% of all pregnant females Large Swedish study looking at pregnancy registry, unblinded n = 500 with SSRI use, 39,000 controls 2.6/2.1 OR elevated risk for PPH/PP anemia 484 mL mean blood loss with SSRI vs 398 mL without SSRI Clinical Implications: No overwhelmingly convincing evidence available as of yet Given bleeding state of pregnancy as well as predilection of SSRIs for bleeding, risk/benefits should be weighed  GI Bleeding
Clinical Implications: Although flawed, pathophysiology and studies combine for reasonable mechanism. Likely should be avoided if possible. PPI Therapy:
 CVA Risks:
Clinical Implications:
Summary:
Do we need to consider bleeding risk when prescribing SSRIs?
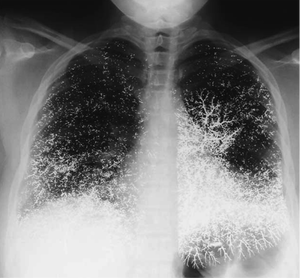
Elemental Mercury – used for home metallurgy, gold smithing, thermometers
Guiterrez, F. Leon, L. Elemental Mercury Embolism to the Lung. N Engle J Med 2000; 342: 1791. June 15, 2000.  Inorganic Mercury
Management
Antidotes – Chelation Therapy!
Dimercaprol (BAL) – Acute elemental and inorganic mercury
References 1. Aaseth J, Skaug MA, Cao Y, Andersen O. Chelation in metal intoxication-Principles and paradigms. J Trace Elem Med Biol. 2014 2. Cao Y, Skaug MA, Andersen O, Aaseth J. Chelation therapy in intoxications with mercury, lead and copper. J Trace Elem Med Biol. 2014; 3. George GN, Prince RC, Gailer J, et al. Mercury binding to the chelation therapy agents DMSA and DMPS and the rational design of custom chelators for mercury. Chem Res Toxicol. 2004;17(8):999-1006. 4. Sue Y. Chapter 96. Mercury. In: Nelson LS, Lewin NA, Howland M, Hoffman RS, Goldfrank LR, Flomenbaum NE. Eds. Goldfrank’s Toxicologic Emergencies, 9e. New York, NY: McGraw-Hill 2011. 5. Guiterrez, F. Leon, L. Elemental Mercury Embolism to the Lung. N Engl J Med 2000; 342: 1791. June 15, 2000. 6. Aaseth J, Jacobsen D, Andersen O, Wickstrøm E. Treatment of mercury and lead poisonings with dimercaptosuccinic acid and sodium dimercaptopropanesulfonate. A review. Analyst. 1995;120(3):853-4. 7. Aposhian HV, Maiorino RM, Gonzalez-ramirez D, et al. Mobilization of heavy metals by newer, therapeutically useful chelating agents. Toxicology. 1995;97(1-3):23-38. 8. Aposhian HV, Bruce DC, Alter W, Dart RC, Hurlbut KM, Aposhian MM. Urinary mercury after administration of 2,3-dimercaptopropane-1-sulfonic acid: correlation with dental amalgam score. FASEB J. 1992;6(7):2472-6. 9. Aaseth J, Skaug MA, Cao Y, Andersen O. Chelation in metal intoxication-Principles and paradigms. J Trace Elem Med Biol. 2014; 10. Cao Y, Skaug MA, Andersen O, Aaseth J. Chelation therapy in intoxications with mercury, lead and copper. J Trace Elem Med Biol. 2014; 11. Nielsen JB, Andersen O. Effect of four thiol-containing chelators on disposition of orally administered mercuric chloride. Hum Exp Toxicol. 1991; 10: 423-430. 12. Planas-bohne F. The effect of 2,3-dimercaptorpropane-1-sulfonate and dimercaptosuccinic acid on the distribution and excretion of mercuric chloride in rats. Toxicology. 1981;19(3):275-8. 13. Clarkson TW, Magos L, Cox C, et al. Tests of efficacy of antidotes for removal of methylmercury in human poisoning during the Iraq outbreak. J Pharmacol Exp Ther. 1981;218(1):74-83. 14. Ruha AM. Recommendations for provoked challenge urine testing. J Med Toxicol. 2013;9(4):318-25. 15. Bates N. Metallic and inorganic mercury poisoning. Emerg Nurse. 2003;11(1):25-31. 16. Guha mazumder DN, De BK, Santra A, et al. Randomized placebo-controlled trial of 2,3-dimercapto-1-propanesulfonate (DMPS) in therapy of chronic arsenicosis due to drinking arsenic-contaminated water. J Toxicol Clin Toxicol. 2001;39(7):665-74. 17. Muran PJ. Mercury elimination with oral DMPS, DMSA, vitamin C, and glutathione: an observational clinical review. Altern Ther Health Med. 2006;12(3):70-5. 18. .Rooney JP. The role of thiols, dithiols, nutritional factors and interacting ligands in the toxicology of mercury. Toxicology. 2007;234(3):145-56. |
Toxicology BlogAuthorEM Rotators on Toxicology Selected by Feedspot as one of the Top 20 Toxicology Blogs on the web
Archives
March 2018
Categories
All
Disclaimer: All images included on this blog are the sole property of CMC EM Residency and cannot be used or reproduced without written permission. Patient identifiers have been redacted/changed or patient consent has been obtained. Information contained in this blog is the opinion of the author and application of material contained in this blog is at the discretion of the practitioner to verify for accuracy.
|







 RSS Feed
RSS Feed
