|
Authored by: Dr. Chrissy Zauner
Between January 1st-January 22nd, there were 86 cases of intentional ingestion. As of Saturday, February 10, 2018 a YouTube search produces 81,200 video results, both of videos performing the challenge and those against it. YouTube recently announced it would be pulling videos due to the dangers of exposure. Pharmocology/Pharmokinetics: Unclear overall
Treatment:
References:
0 Comments
Authored by Dr. Enola Okonkwo Introduction Artemether Lumefantrine is an oral medication which is used in treatment of chloroquine resistant uncomplicated malaria. Artemether Lumefantrine falls within the drug class of Artemisinin-based combination therapies which have been found to be highly effective in treating malaria and are substantially less toxic than quinoline antimalarials. Artemisins are derived from a Chinese medicinal herb which comes from the sweet wormwood plant (1). Artemether Lumefantrine, known by the brand name Coartem, is the most widely used artemisinin based combination therapy used in Africa and is one of three medications recommended by the CDC as first line oral treatment for uncomplicated falciparum malaria within the United States (2). Structure 1 tablet = 20mg artemether and 120 mg lumefantrine.
Dosing is weight based. For patients > 35 kg they are taking 4 tablets per dose twice daily for 3 days. Pharmacology/Pharmacokinetics: Absorption/Distribution/Metabolism/Excretion Artemeter is rapidly absorbed. Peak plasma level of artemeter is approximately 2 hours. The half-life after ingestion is 2 hours (4-5). 95% of artemether is protein bound but quickly undergoes metabolism to dihydroartemisin which is approximately 50% protein bound. Half life of dihydroartemisin is also around 2 hours (5). Bioavailability increased 2-3 fold when consumed with food (4). Lumefantrine – Initial absorption at approximately 2 hours. Peak plasma level approximately 6-8 hours. Half life is 4-5 days. It is highly protein bound and has a large volume of distribution. High fat meals significantly increase absorption 16 fold (4). Significant Drug/Drug interactions Numerous theoretic drug interactions have been reported. However, there is scant data available reporting clinical effects of drug interactions. -May enhance QTc prolonging effects -May enhance toxic effects of other antimalarial or HIV medications -May increase serum concentration of antipsychotics -May decrease serum concentration of estrogen related contraceptives -May decrease serum concentration of CYP3A4 Substrates Metabolic Pathways and active metabolites Arthemeter is metabolized by the liver to the active metabolite dihydroartemisinin primarily by CYP3A4/5. Lumefantrine is metabolized by the liver to desbutyl-lumefantrine by CYP3A4. Studies suggest that artemisins inhibit the sarco/endoplasmic reticulum Ca ATPases of the malaria parasite causing rapid reduction in the parasite (6-7). The mechanism of lumefantrine is unknown but both agents inhibit nucleic acid and protein synthesis. Artemether rapidly kills the majority of the parasite and lumefantrine goes on to kill the residual remaining parasite (4-5). Mechanism of Toxicity Artemether Lumefantrine has been extensively used for the treatment of malaria and appears to have an excellent safety profile. There are no reported overdoses or intentional ingestions. There has been no evidence of significant systemic or local toxicity reported in any large human study (1, 20-21). However, there is animal data and a few small human studies and case reports which suggest neurotoxicity and QT prolongation are possible toxic effects of artimisinins (14). Neurotoxicity was especially common in animal studies both clinically and histologically (8-10, 14). In vitro studies have postulated that neurotoxicity results secondary to ATP depletion of neurons and free radical generation created by the breakdown of artemisins (Schmuck 2002, Smith 1998) . Toover et. el wrote a review article cautioning that neurotoxic effects may also occur in humans more frequently than previously thought, though many critics viewed their review as bias (8, 13). There are a small number of case reports within humans suggesting that artemisins contributed to neurologic symptoms such as anxiety, tremor, and ataxia (8, 15-16). A small study looking at construction workers treated with arthemeter suggested that individuals had hearing loss following the administration of artemether (17). Obviously, this study is limited by a serious potential confounder and attempts to reproduce these results have failed (18). One case report of delayed hemolytic anemia, not thought to be related to malaria was reported (19). Clinical Toxicity Given patients taking artemether lumefantrine have malaria, it is difficult to distinguish between symptoms caused by the disease state of malaria versus symptoms which may be attributed to medication toxicity. Clinical toxicity appears to be very rare or possibly underreported. A large review of 188 studies including over 9,000 patients found no serious clinical toxicity (21). This review article used methodology similar to Cochrane review and reported transient neutropenia in 1.3%, reticulocytopenia (0.6%), elevated liver enzymes (0.9%), and transient bradycardia and prolonged QT (1.1%). The most common side effects reported were gastrointestinal. The authors conclude that Artemether Lumefantrine is a safe and efficacious drug. Common adverse events (4, 20-21) Abdominal pain Anorexia Vomiting Diarrhea Headache dizziness. Pruritis and Rash < 2% Laboratories No specific toxicology labs are available Management of Toxicity No accepted recommendations regarding toxicity given the scarcity of data suggesting toxicity. Below are general recommendations based off of literature review: Avoid concurrent use of additional antimalarial medications Use caution administering additional drugs which can prolong QT Use caution in hypokalemia and replete electrolytes as needed Use caution in patients with prolonged QT Obtain EKG as screening for arrhythmia References: 1. Hien, T. T., White, N. J., & White. (1993). Qinghaosu. The Lancet, 341(8845), 603–608. https://doi.org/10.1016/0140-6736(93)90362-K 2. Center for Disease Control. (2009). Guidelines for treatment of malaria in the United States. Treatment Table Update, May, (770), 855–857. Retrieved from http://scholar.google.com/scholar?hl=en&btnG=Search&q=intitle:Guidelines+for+Treatment+of+Malaria+in+the+United+States#1 3. Amin, N. C., Fabre, H., Blanchin, M.-D., Montels, J., & Aké, M. (2013). Determination of artemether and lumefantrine in anti-malarial fixed-dose combination tablets by microemulsion electrokinetic chromatography with short-end injection procedure. Malaria Journal, 12, 202. https://doi.org/10.1186/1475-2875-12-202 4. Waning, B., & Montange, M. (2015). Access Pharmacy. Retrieved from http://accesspharmacy.mhmedical.com/content.aspx?bookid=438§ionid=40428529 5. White, N. J., Van Vugt, M., & Ezzet, F. (1999). Clinical pharmacokinetics and pharmacodynamics of artemether-lumefantrine. Clinical Pharmacokinetics. Adis International Ltd. https://doi.org/10.2165/00003088-199937020-00002 6. Krishna, S., Pulcini, S., Moore, C. M., Teo, B. H. Y., & Staines, H. M. (2014, January). Pumped up: Reflections on PfATP6 as the target for artemisinins. Trends in Pharmacological Sciences. https://doi.org/10.1016/j.tips.2013.10.007 7. Eckstein-Ludwig, U., Webb, R. J., Van Goethem, I. D. A., East, J. M., Lee, A. G., Kimura, M., … Krishna, S. (2003). Artemisinins target the SERCA of Plasmodium falciparum. Nature, 424(6951), 957–961. https://doi.org/10.1038/nature01813 8. Toovey, S. (2006, October 10). Are currently deployed artemisinins neurotoxic? Toxicology Letters. https://doi.org/10.1016/j.toxlet.2006.06.001 9. Genovese, R. F., Newman, D. B., Li, Q., Peggins, J. O., & Brewer, T. G. (1998). Dose-dependent brainstem neuropathology following repeated arteether administration in rats. Brain Research Bulletin, 45(2), 199–202. https://doi.org/10.1016/S0361-9230(97)00339-0 10. Petras, J. M., Young, G. D., Bauman, R. A., Kyle, D. E., Gettayacamin, M., Webster, H. K., … Brewer, T. G. (2000). Arteether-induced brain injury in Macaca mulatta. I. The precerebellar nuclei: The lateral reticular nuclei, paramedian reticular nuclei, and perihypoglossal nuclei. Anatomy and Embryology, 201(5), 383–397. https://doi.org/10.1007/s004290050326 11. Schmuck, G., Roehrdanz, E., Haynes, R. K., & Kahl, R. (2002). Neurotoxic mode of action of artemisinin. Antimicrobial Agents and Chemotherapy, 46(3), 821–827. https://doi.org/10.1128/AAC.46.3.821-827.2002 12. Smith, S. L., Maggs, J. L., Edwards, G., Ward, S. A., Park, B. K., & McLean, W. G. (1998). The role of iron in neurotoxicity: a study of novel antimalarial drugs. Neurotoxicology, 19(4–5), 557–559. 13. Toovey, S. (2006, October 10). Are currently deployed artemisinins neurotoxic? Toxicology Letters. https://doi.org/10.1016/j.toxlet.2006.06.001 14. Brewer, T. G., Peggins, J. O., Grate, S. J., Petras, J. M., Levine, B. S., Weina, P. J., … Schuster, B. G. (1994). Neurotoxicity in animals due to arteether and artemether. Transactions of the Royal Society of Tropical Medicine and Hygiene, 88, 33–36. https://doi.org/10.1016/0035-9203(94)90469-3 15. Miller, L. G., & Panosian, C. B. (1997). Ataxia and slurred speech after artesunate treatment for falciparum malaria. New England Journal of Medicine, 336(18), 1328. https://doi.org/10.1056/NEJM199705013361818 16. Franco-Paredes, C., Dismukes, R., Nicolls, D., & Kozarsky, P. E. . (2005). Neurotoxicity Due to Antimalarial Therapy Associated with Misdiagnosis of Malaria. Clinical Infectious Diseases : An Official Publication of the Infectious Diseases Society of America, 40(11), 1710–1711. https://doi.org/10.1086/430180 17. Toovey, S. (2006). A case-control auditory evaluation of patients treated with artemether-lumefantrine. American Journal of Tropical Medicine and Hygeine 74, 939-940. 18. Hutagalung, R., Htoo, H., Nwee, P., Arunkamomkiri, J., Zwang, J., Carrara, V. I., … Nosten, F. (2006). A case-control auditory evaluation of patients treated with artemether-lumefantrine. American Journal of Tropical Medicine and Hygiene, 74(2), 211–214. https://doi.org/74/2/211 [pii] 19. Hasegawa, C., Kudo, M., Maruyama, H., Kimura, M. (2017). Severe delayed haemolytic anaemia associated with artemether-lumefantrine treatment of malaria in a Japanese traveler. Journal of Infection and Chemotherapy. In press. https://doi.org/10.1016/j.jiac.2017.10.008 20. Alkadi, H. O. (2007, November). Antimalarial drug toxicity: A review. Chemotherapy. https://doi.org/10.1159/000109767 21. Ribeiro, I. R., & Olliaro, P. (1998). Safety of artemisinin and its derivatives. A review of published and unpublished clinical trials. Medecine Tropicale : Revue Du Corps de Sante Colonial, 58(3 Suppl), 50–53. Retrieved from http://ovidsp.ovid.com/ovidweb.cgi?T=JS&PAGE=reference&D=med4&NEWS=N&AN=10212898 Authored by Dr. Kyle Roedersheimer Introduction to ADHD
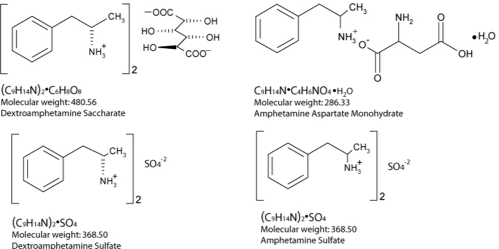
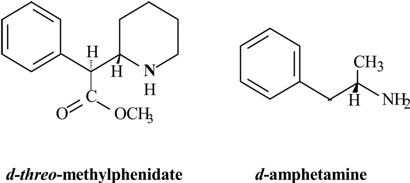
History of Amphetamines
Psychostimulants
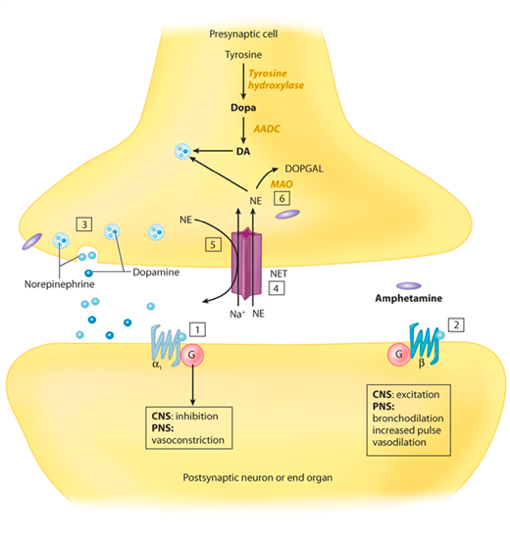
Mechanism of action
Pharmacology/Pharmacokinetics:
Therapeutic levels
Significant Drug/Drug Interactions
Metabolic Pathways and active metabolites
Diagnostic Testing and Laboratories
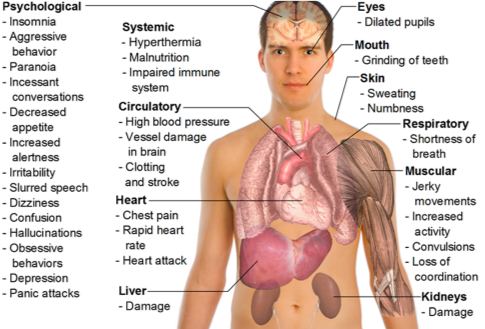
Treatment/Management
What is Stuttering? What causes it?
Multiple Case Reports Discussing Stuttering Associated with ADHD Medications
References
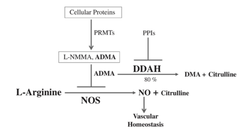
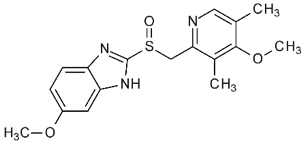
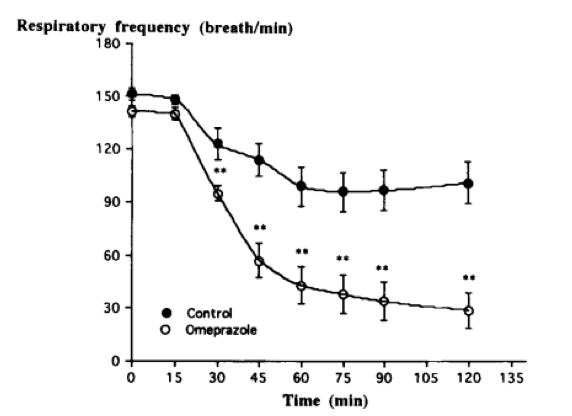
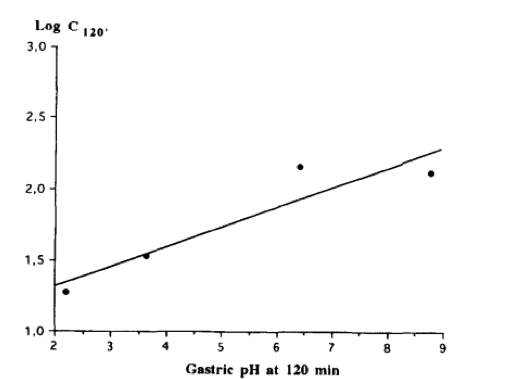
Authored By: Meredith Binford Background: Proton pump inhibitors (PPIs) inhibit H+K+ATPase proton pumps in the stomach to reduce acid excretion. Among the most frequently prescribed drugs worldwide and are generally well tolerated. Now they are available for long-term use without medical supervision. Often prescribed to circumvent GI side effects from dual antiplatelet agents used for cardiovascular disease secondary prevention. However recent observational studies have linked PPI use to increased cardiovascular risk (MI and stroke) even in patients not taking antiplatelet agents. Mechanism: -impair lysosomal acidification and enzyme activity -accelerate telomere erosion -speed endothelial aging as manifested by impaired cell perforation and angiogenesis -impair the NO synthase pathway though asymmetric dimethylarginine (ADMA) 3 recent articles investigate PPIs and risk of stroke: 1. A large retrospective study from the Taiwan National Health Insurance Database studied whether PPI use alone could be associated with first time ischemic stroke. In a propensity score matched analysis (400,000 patients) over 120 days PPI use was associated with a higher risk of hospitalization due to ischemic stroke with hazard ratio of 1.36 (95% CI 1.14-1.62, P=0.001). In subgroup analysis, the risk was more prominent in those less than 60 years of age but gender, prior MI, diabetes, hypertension, use of antiplatelet agents or NSAIDs, or types of PPI did not have an influence on the risk. In the nested case-control analysis (30,000 patients), the adjusted odds ratios for PPI use were 1.77 (95% CI 1.45-2.17, P<0.001) within 30 days and 1.65 (95% CI 1.31-2.08, P<0.001) 31 to 90 days before ischemic stroke. The proportions of H2 blocker users and non users were comparable before the event. One of the major arguments against the association is confounding by indication for PPI use as PPIs are used over H2 blockers in the presence of more severe acid related GI disorders. Other uncorrected confounders include obesity, lack of exercise, smoking, compliance with hypertension treatment, diabetes, hyperlipidemia, other medical illnesses. Also limited by 120 day follow up which may have masked confounders of PPI use. 2. Danish registry of people who underwent elective EGD (215,000 patients) was analyzed in a time varying design to asses dose response effects on hazard ratios as well as in a baseline treatment design to show impact of long term use on risks of first time stroke and MI. Current PPI exposure was associated with significantly higher rates of both ischemic stroke with hazard ratio of 1.13 (95% CI 1.08-1.19) and MI with hazard ratio of 1.31 (95% CI 1.23-1.39) after adjusting for age, sex, comorbidities and concomitant medications. High dose PPI was associated with increased rates of ischemic stroke with hazard ratio of 1.31 (95% CI 1.21-1.42) and MI with hazard ratio of 1.43 (95% CI 1.30-1.57). H2 blockers were not significantly associated with ischemic stroke or MI. Long-term PPI users had a 29% greater absolute risk of ischemic stroke and a 36% greater absolute risk of MI within six months period. One limitation is confounding that PPI identifies unhealthy individuals. When estimating impact of smoking habit, obesity, and exercise the net confounding bias was between 3.8 and 4.1% which reduced the hazard ratio to 1.08 for ischemic stroke and 1.26 for MI. Also concern of a lead in bias as subjects were likely exposed to PPI therapy before EGD. 3. Analysis from the Nurses Health Study and the Health Professionals Follow Up Study (950,000 person years) observed an association between regular PPI use and risk of ischemic stroke in age-adjusted models and multivariate models adjusting for known risk factors for ischemic stroke that was consistent with findings of prior reports. However additional adjustment for factors that may be associated with initiation and continuation of PPI therapy including history of peptic ulcer disease, GERD, GI bleeding and prior H2 blocker use substantially attenuated the association. They found that participants who were regular PPI users at baseline tended to have higher rates of chronic medical conditions including prior history of hypertension, hyperlipidemia, CAD, diabetes, as well as lower levels of physical activity expenditure. Among women, regular PPI users were more likely to have a higher BMI and a prior history of menopausal hormonal use. Association of PPI use with ischemic stroke may be due to residual confounding by factors associated with the indication for PPI use, for example metabolic syndrome. Conclusion: There is a plausible biological mechanism to explain the association of PPIs with increased cardiovascular risk. Large retrospective studies show association especially with longer use and higher dose PPI. However there is concern of confounding as may be related to more unhealthy patients using PPIs. References: Sehested, TSG et al. Long term use of proton pump inhibitors, dose-response relationship and associated risk of scheming stroke and myocardial infarction. Journal Internal Medicine 2017, doi: 10.1111/joim.12698. Nguyen LH, Lochhead P, Joshi AD, Cao Y, Ma W, Khalili H, Rimm EB, Rexrode KN< Chan AT. No significant association between proton pump inhibitor use and risk of stroke after adjustment for lifestyle factors and indication. Gastroenterology 2018, doi: 10.1053/j.gastro.2017.12.006. Wang Y-F, Chen Y-T, Luo J-C, Chen T-J, Wu J-C, Wang S- J. Proton-pump inhibitor use and the risk of first-time ischemic stroke in the general population: a nationwide population-based study. Am J Gastroenterol 2017; 112: 1– 10. Yepuri G, Sukhovershin R, Nazari-Shafti TZ, Petrascheck M, Ghebre YT, Cooke JP. Proton pump inhibitors accel- erate endothelial senescence. Circ Res 2016; 118: e36–42. Ghebremariam YT, Lependu P, Lee JC et al. Unexpected effect of proton pump inhibitors: elevation of the cardiovascular risk factor asymmetric dimethylarginine. Circulation 2013; 128: 845–53. Cooke JP. Asymmetrical dimethylarginine: the u€ber marker? Circulation 2004; 109: 1813–9. Authored by Dr. Natalie Wood Uses: -The flesh of the fruit consists of an edible, white pulp , some fiber, and a core of indigestible, black seeds. Used in foods -eaten as dessert -smoothies, fruit juice drinks -candy -sorbets and ice cream flavoring -Traditional medicinal uses for diarrhea, liver disease, heart disease, intestinal parasites, diabetes, and cancer. -Pesticides, insecticides and topical insect repellant in latin america -shampoos for treating lice and ticks
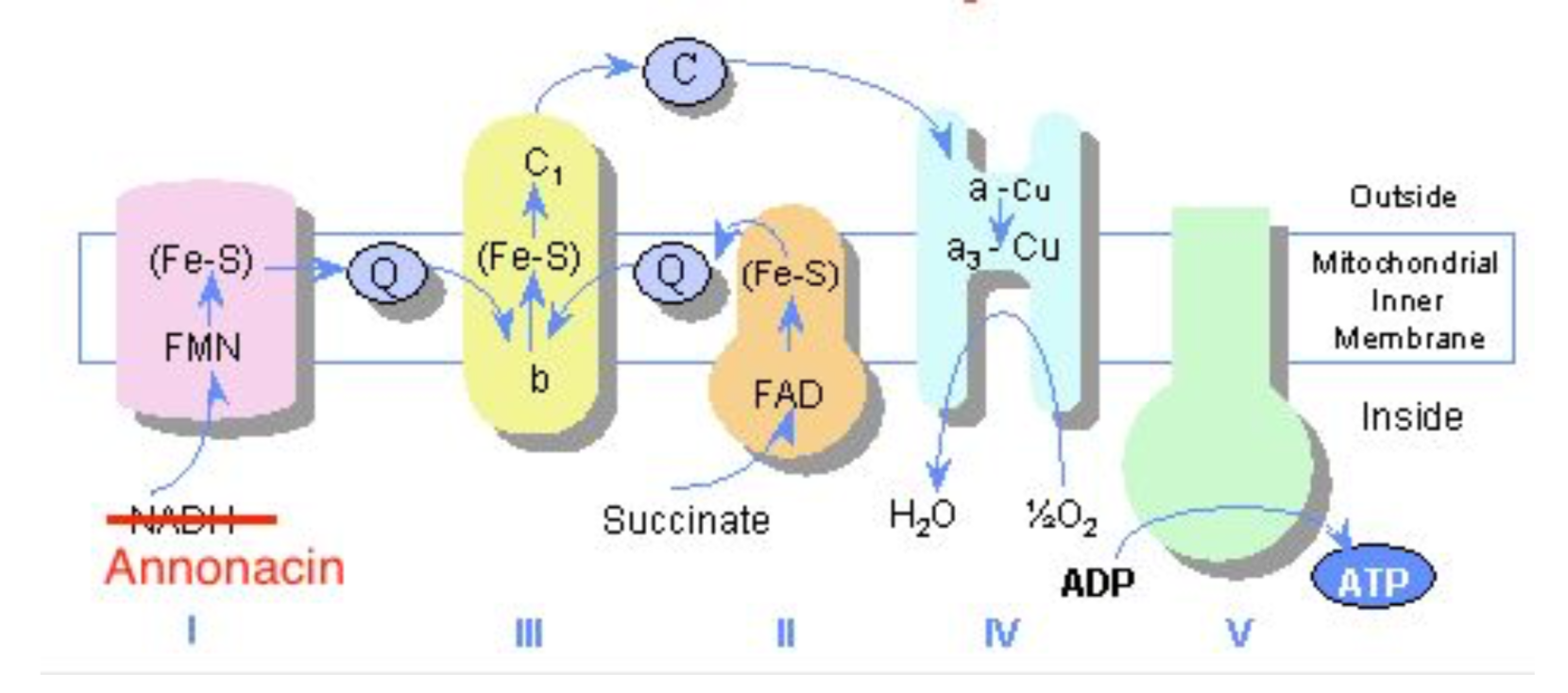
-This is exceptionally frequent compared to epidemiological data from European countries where atypical parkinsonism accounts for only approximately 5% of all cases - A higher proportion of patients with atypical parkinsonism (consumed soursop fruit 97%; consumed herbal tea 83%) than patients with Parkinson’s disease (fruit 59%; herbal tea 18%) or control with no Parkinson symptoms (fruit 60%; herbal tea 43%). -When directly injecting rats for one month with the estimated amounts of annonacin ingested in a year by eating one fruit daily, this induced neurodegeneration in the basal ganglia and mesencephalon (Champy et al., 2004) -As an indication of its potential toxicity, an adult who consumes one fruit or can of nectar a day is estimated to ingest over 1 year the amount of annonacin that induced brain lesions in rats receiving purified annonacin by intravenous infusion. -Multiple studies showing toxicity in vitro to dopaminergic neurons -Cultured neurons treated with low doses of annonacin exhibit hallmarks of tauopathy, including increased tau concentration, tau hyperphosphorylation, cell death and somatodendritic redistribution of hyperphosphorylated tau which is that same pathophysiology of progressive supranuclear palsy. -Studied as a cytotoxic agent for use in cancer chemotherapy -Extract from the seeds and leaves have IC50 comparable to doxorubicin at .57μg/mL (seed) and .36μg/mL (leaves) compared to .11μg/mL (doxorubicin) in leukemia cells. -Mechanism of action is via annonacin, the acetogenin causing mitochondrial membrane potential disruption. It inhibits NADH dehydrogenase on Complex I of the electron transport chain. Creates reactive O2 species that can damage DNA and other components of mitochondria -Annona muricata leaf extract reduced the tumor's size and weight, showed anti-metastatic features, and induced apoptosis in vitro and in vivo of the 4 T1 breast cancer cell lines. Furthermore, it decreased the level of nitric oxide and malondialdehyde in tumor while also increased the level of white blood cell, T-cell, and natural killer cell population. Pharmacology -LD 50 >2g/kg -In IV administration to Rats 3800 and 7600 μg/kg daily caused neurodegeneration over a period of 28 days -it appears that chronic consumption causes the neurodegeneration Presentation -Levodopa resistant parkinsonism similar to progressive supranuclear palsy -PSP presents with loss of balance, bradykinesia, rigidity, problems controlling eye movements, issues with speech and swallowing -Patients in Guadaloupe: -early postural instability -supranuclear oculomotor dysfunction -greater frequency of patients with tremor as compared to typical PSP -greater frequency of patients with hallucinations as compared to typical PSP Laboratories -no laboratory evidence of annonacin toxicity -annonacin can be identified in brain neurons on autopsy Treatment/Management -stop ingestion of soursop -poor response to levodopa or other parkinson’s treatment agents Resources Caparros-Lefebvre, Dominique, Nicolas Sergeant, Andrew Lees, Agnes Camuzat, Susan Daniel, Annie Lannuzel, Alexis Brice, Eduardo Tolosa, Andre Delacourte, and Charles Duyckaerts. "Guadeloupean parkinsonism: a cluster of progressive supranuclear palsy-like tauopathy." Brain 125.4 (2002): 801-11. Web. 9 July 2017. Champy, Pierre, Alice Melot, Vincent Guérineau Eng, Christophe Gleye, Djibril Fall, Gunter U. Höglinger, Merle Ruberg, Annie Lannuzel, Olivier Laprévote, Alain Laurens, and Reynald Hocquemiller. "Quantification of acetogenins inAnnona muricata linked to atypical parkinsonism in guadeloupe." Movement Disorders 20.12 (2005): 1629-633. Web. 9 July 2017. Clement, Yuri N., Varune Mahase, Annelise Jagroop, Kelly Kissoon, Aarti Maharaj, Prashant Mathura, Chrys Mc Quan, Divya Ramadhin, and Cherrista Mohammed. "Herbal remedies and functional foods used by cancer patients attending specialty oncology clinics in Trinidad." BMC Complementary and Alternative Medicine 16.1 (2016): n. pag. Web. 09 July 2017. Coria-Téllez, Ana V., Efigenia Montalvo-Gónzalez, Elhadi M. Yahia, and Eva N. Obledo-Vázquez. "Annona muricata: A comprehensive review on its traditional medicinal uses, phytochemicals, pharmacological activities, mechanisms of action and toxicity." Arabian Journal of Chemistry (2016): n. pag. Web. 9 July 2017. Esposti, M. Degli, A. Ghelli, M. Ratta, D. Cortes, and E. Estornell. "Natural substances (acetogenins) from the familyAnnonaceaeare powerful inhibitors of mitochondrial NADH dehydrogenase (Complex I)." Biochemical Journal 301.1 (1994): 161-67. Web. 9 July 2017. Kuete, Victor, Joachim K. Dzotam, Igor K. Voukeng, Aimé G. Fankam, and Thomas Efferth. "Cytotoxicity of methanol extracts of Annona muricata, Passiflora edulis and nine other Cameroonian medicinal plants towards multi-factorial drug-resistant cancer cell lines." SpringerPlus 5.1 (2016): n. pag. Web. Salama, Mohamed, and Oscar Arias-Carrión. "Natural toxins implicated in the development of Parkinson’s disease." Therapeutic Advances in Neurological Disorders 4.6 (2011): 361-73. Web. 9 July 2017. Lannuzel, A., G. U. Hoglinger, S. Verhaeghe, L. Gire, S. Belson, M. Escobar-Khondiker, P. Poullain, W. H. Oertel, E. C. Hirsch, B. Dubois, and M. Ruberg. "Atypical parkinsonism in Guadeloupe: a common risk factor for two closely related phenotypes?" Brain 130.3 (2007): 816-27. Web. 9 July 2017. Soursop . N.p., n.d. Web. 09 July 2017.
|
Toxicology BlogAuthorEM Rotators on Toxicology Selected by Feedspot as one of the Top 20 Toxicology Blogs on the web
Archives
March 2018
Categories
All
Disclaimer: All images included on this blog are the sole property of CMC EM Residency and cannot be used or reproduced without written permission. Patient identifiers have been redacted/changed or patient consent has been obtained. Information contained in this blog is the opinion of the author and application of material contained in this blog is at the discretion of the practitioner to verify for accuracy.
|









 RSS Feed
RSS Feed
